Inhibitory effects of SKF96365 on the activities of K(+) channels in mouse small intestinal smooth muscle cells
- PMID: 26498720
- PMCID: PMC4785108
- DOI: 10.1292/jvms.15-0346
Inhibitory effects of SKF96365 on the activities of K(+) channels in mouse small intestinal smooth muscle cells
Abstract
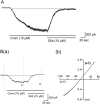
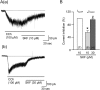
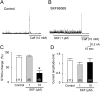
In order to investigate the effects of SKF96365 (SKF), which is a non-selective cationic channel blocker, on K(+) channel currents, we recorded currents through ATP sensitive K(+) (IKATP), voltage-gated K(+) (IKv) and Ca(2+) activated K(+) channels (IBK) in the absence and presence of SKF in single small intestinal myocytes of mice with patch-clamp techniques. SKF (10 µM) reversibly abolished IKATP that was induced by cromakalim (10 µM), which is a selective ATP sensitive K(+) channel opener. These inhibitory effects were induced in a concentration-dependent and voltage-independent manner. The 50% inhibitory concentration (IC50) was 0.85 µM, which was obviously lower than that reported for the muscarinic cationic current. In addition, SKF (1 µM ≈ the IC50 value in IKATP suppression) reversibly inhibited the IKv that was induced by repetitive depolarizing pulses from -80 to 20 mV. However, the extent of the inhibitory effects was only ~30%. In contrast, SKF (1 µM) had no significant effects on spontaneous transient IBK and caffeine-induced IBK. These results indicated that SKF inhibited ATP sensitive K(+) channels and voltage-gated K(+) channels, with the ATP sensitive K(+) channels being more sensitive than the voltage-gated K(+) channels. These inhibitory effects on K(+) channels should be considered when SKF is used as a cationic channel blocker.
Figures


References
-
- Aguilar-Bryan L., Clement J. P., 4th, Gonzalez G., Kunjilwar K., Babenko A., Bryan J.1998. Toward understanding the assembly and structure of KATP channels. Physiol. Rev. 78: 227–245. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

