Randomized Trial of Late Surfactant Treatment in Ventilated Preterm Infants Receiving Inhaled Nitric Oxide
- PMID: 26500107
- PMCID: PMC4698022
- DOI: 10.1016/j.jpeds.2015.09.031
Randomized Trial of Late Surfactant Treatment in Ventilated Preterm Infants Receiving Inhaled Nitric Oxide
Abstract
Objective: To assess whether late surfactant treatment in extremely low gestational age (GA) newborn infants requiring ventilation at 7-14 days, who often have surfactant deficiency and dysfunction, safely improves survival without bronchopulmonary dysplasia (BPD).
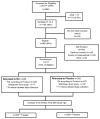
Study design: Extremely low GA newborn infants (GA ≤28 0/7 weeks) who required mechanical ventilation at 7-14 days were enrolled in a randomized, masked controlled trial at 25 US centers. All infants received inhaled nitric oxide and either surfactant (calfactant/Infasurf) or sham instillation every 1-3 days to a maximum of 5 doses while intubated. The primary outcome was survival at 36 weeks postmenstrual age (PMA) without BPD, as evaluated by physiological oxygen/flow reduction.
Results: A total of 511 infants were enrolled between January 2010 and September 2013. There were no differences between the treated and control groups in mean birth weight (701 ± 164 g), GA (25.2 ± 1.2 weeks), percentage born at GA <26 weeks (70.6%), race, sex, severity of lung disease at enrollment, or comorbidities of prematurity. Survival without BPD did not differ between the treated and control groups at 36 weeks PMA (31.3% vs 31.7%; relative benefit, 0.98; 95% CI, 0.75-1.28; P = .89) or 40 weeks PMA (58.7% vs 54.1%; relative benefit, 1.08; 95% CI, 0.92-1.27; P = .33). There were no between-group differences in serious adverse events, comorbidities of prematurity, or severity of lung disease to 36 weeks.
Conclusion: Late treatment with up to 5 doses of surfactant in ventilated premature infants receiving inhaled nitric oxide was well tolerated, but did not improve survival without BPD at 36 or 40 weeks. Pulmonary and neurodevelopmental assessments are ongoing.
Trial registration: ClinicalTrials.gov: NCT01022580.
Copyright © 2016 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
-
- Northway WH, Jr, Rosan RC, Porter DY. Pulmonary disease following respirator therapy of hyaline-membrane disease. Bronchopulmonary dysplasia. N Engl J Med. 1967;276:357–68. - PubMed
-
- Ehrenkranz RA, Walsh MC, Vohr BR, Jobe AH, Wright LL, Fanaroff AA, et al. Validation of the National Institutes of Health consensus definition of bronchopulmonary dysplasia. Pediatrics. 2005;116:1353–60. - PubMed
-
- Hennessy EM, Bracewell MA, Wood N, Wolke D, Costeloe K, Gibson A, et al. Respiratory health in pre-school and school age children following extremely preterm birth. Archives of disease in childhood. 2008;93:1037–43. - PubMed
-
- Husain AN, Siddiqui NH, Stocker JT. Pathology of arrested acinar development in postsurfactant bronchopulmonary dysplasia. Human pathology. 1998;29:710–7. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical