Revisiting Plant Plasma Membrane Lipids in Tobacco: A Focus on Sphingolipids
- PMID: 26518342
- PMCID: PMC4704565
- DOI: 10.1104/pp.15.00564
Revisiting Plant Plasma Membrane Lipids in Tobacco: A Focus on Sphingolipids
Abstract
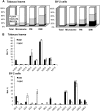
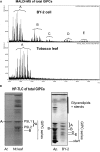
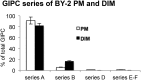
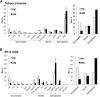
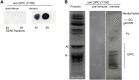
The lipid composition of plasma membrane (PM) and the corresponding detergent-insoluble membrane (DIM) fraction were analyzed with a specific focus on highly polar sphingolipids, so-called glycosyl inositol phosphorylceramides (GIPCs). Using tobacco (Nicotiana tabacum) 'Bright Yellow 2' cell suspension and leaves, evidence is provided that GIPCs represent up to 40 mol % of the PM lipids. Comparative analysis of DIMs with the PM showed an enrichment of 2-hydroxylated very-long-chain fatty acid-containing GIPCs and polyglycosylated GIPCs in the DIMs. Purified antibodies raised against these GIPCs were further used for immunogold-electron microscopy strategy, revealing the distribution of polyglycosylated GIPCs in domains of 35 ± 7 nm in the plane of the PM. Biophysical studies also showed strong interactions between GIPCs and sterols and suggested a role for very-long-chain fatty acids in the interdigitation between the two PM-composing monolayers. The ins and outs of lipid asymmetry, raft formation, and interdigitation in plant membrane biology are finally discussed.
© 2016 American Society of Plant Biologists. All Rights Reserved.
Figures


 , where A is the mean molecular area, X is the molar fraction, subscripts 1 and 2 refer to pure components 1 and 2, respectively, and subscript refers 12 to their mixtures; and
, where A is the mean molecular area, X is the molar fraction, subscripts 1 and 2 refer to pure components 1 and 2, respectively, and subscript refers 12 to their mixtures; and , where ΔGid is the free energy for ideal mixing and can be calculated from the following equation:
, where ΔGid is the free energy for ideal mixing and can be calculated from the following equation:  , where R is the universal gas constant and T is the absolute temperature.
, where R is the universal gas constant and T is the absolute temperature.

References
-
- Blaas N, Humpf HU (2013) Structural profiling and quantitation of glycosyl inositol phosphoceramides in plants with Fourier transform mass spectrometry. J Agric Food Chem 61: 4257–4269 - PubMed
-
- Brasseur R. (1990). TAMMO: theoretical analysis of membrane molecular organisation. In Molecular Description of Biological Membrane Components by Computer-Aided Conformational Analysis. CRC Press, Boca Raton, FL, pp 203–219
-
- Buré C, Cacas JL, Wang F, Gaudin K, Domergue F, Mongrand S, Schmitter JM (2011) Fast screening of highly glycosylated plant sphingolipids by tandem mass spectrometry. Rapid Commun Mass Spectrom 25: 3131–3145 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

