Effects of long-term resistance exercise training on autophagy in rat skeletal muscle of chloroquine-induced sporadic inclusion body myositis
- PMID: 26525066
- PMCID: PMC4624124
- DOI: 10.5717/jenb.2015.15090710
Effects of long-term resistance exercise training on autophagy in rat skeletal muscle of chloroquine-induced sporadic inclusion body myositis
Abstract
Purpose: We examined whether resistance exercise training restores impaired autophagy functions caused by Chloroquine (CQ)-induced Sporadic Inclusion Body Myositis (sIBM) in rat skeletal muscle.
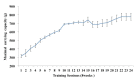
Methods: Male wistar rats were randomly assigned into three groups: Sham (n = 6), CQ (n = 6), and CQ + Exercise (CE, n = 6). To create a rat model of sIBM, rats in the CQ and CE group were intraperitoneally injected with CQ 5 days a week for 16 weeks. Rats in the CE group performed resistance exercise training 3 times a week for 8 weeks in conjunction with CQ starting from week 9 to week 16. During the training period, maximal carrying load, body weight, muscle weight, and relative muscle weight were measured. Autophagy responses were examined by measuring specific markers.
Results: While maximal carrying capacity for resistance exercise training was dramatically increased in the CE group, no significant changes occurred in the skeletal muscle weight as well as in the relative muscle weight of CE compared to the other groups. CQ treatment caused significant increases in the levels of Beclin-1 and p62, and decreases in the levels of LAMP-2 proteins. Interestingly, no significant differences in the LC3-II/I ratio or the LC3-II protein levels were observed. Although CQ-treatment groups suppressed the levels of the potent autophagy inducer, BNIP3, p62 levels were decreased in only the CE group.
Conclusion: Our findings demonstrate that sIBM induced by CQ treatment results in muscle degeneration via impaired autophagy and that resistance exercise training improves movable loading activity. Finally, regular exercise training may provide protection against sIBM by enhancing the autophagy flux through p62 protein.
Keywords: Resistance exercise; autophagy; chloroquine; sIBM; skeletal muscles.
Figures


Similar articles
-
Effect of Resistance Exercise on Muscle Metabolism and Autophagy in sIBM.Med Sci Sports Exerc. 2017 Aug;49(8):1562-1571. doi: 10.1249/MSS.0000000000001286. Med Sci Sports Exerc. 2017. PMID: 28333717
-
Resistance Exercise Improves Mitochondrial Quality Control in a Rat Model of Sporadic Inclusion Body Myositis.Gerontology. 2019;65(3):240-252. doi: 10.1159/000494723. Epub 2019 Jan 14. Gerontology. 2019. PMID: 30641518
-
The immune system in sporadic inclusion body myositis patients is not compromised by blood-flow restricted exercise training.Arthritis Res Ther. 2019 Dec 18;21(1):293. doi: 10.1186/s13075-019-2036-2. Arthritis Res Ther. 2019. PMID: 31852482 Free PMC article. Clinical Trial.
-
Exercise-mediated modulation of autophagy in skeletal muscle.Scand J Med Sci Sports. 2018 Mar;28(3):772-781. doi: 10.1111/sms.12945. Epub 2017 Aug 4. Scand J Med Sci Sports. 2018. PMID: 28685860 Review.
-
A Preclinical Systematic Review of the Effects of Chronic Exercise on Autophagy-Related Proteins in Aging Skeletal Muscle.Front Physiol. 2022 Jul 14;13:930185. doi: 10.3389/fphys.2022.930185. eCollection 2022. Front Physiol. 2022. PMID: 35910582 Free PMC article.
Cited by
-
Sarcopenia targeting with autophagy mechanism by exercise.BMB Rep. 2019 Jan;52(1):64-69. doi: 10.5483/BMBRep.2019.52.1.292. BMB Rep. 2019. PMID: 30526769 Free PMC article. Review.
-
Autophagy-Dependent Beneficial Effects of Exercise.Cold Spring Harb Perspect Med. 2017 Aug 1;7(8):a029777. doi: 10.1101/cshperspect.a029777. Cold Spring Harb Perspect Med. 2017. PMID: 28270532 Free PMC article. Review.
-
Polyamines and Physical Activity in Musculoskeletal Diseases: A Potential Therapeutic Challenge.Int J Mol Sci. 2023 Jun 6;24(12):9798. doi: 10.3390/ijms24129798. Int J Mol Sci. 2023. PMID: 37372945 Free PMC article. Review.
-
Excess Accumulation of Lipid Impairs Insulin Sensitivity in Skeletal Muscle.Int J Mol Sci. 2020 Mar 12;21(6):1949. doi: 10.3390/ijms21061949. Int J Mol Sci. 2020. PMID: 32178449 Free PMC article. Review.
-
Mitochondrial Function and Protein Turnover in the Diaphragm are Altered in LLC Tumor Model of Cancer Cachexia.Int J Mol Sci. 2020 Oct 22;21(21):7841. doi: 10.3390/ijms21217841. Int J Mol Sci. 2020. PMID: 33105841 Free PMC article.
References
-
- Needham M, Corbett A, Day T, Christiansen F, Fabian V, Mastaglia FL. Prevalence of sporadic inclusion body myositis and factors contributing to delayed diagnosis. Journal of clinical neuroscience : official journal of the Neurosurgical Society of Australasia. 2008;15:1350–3. - PubMed
-
- Cox FM, Titulaer MJ, Sont JK, Wintzen AR, Verschuuren JJ, Badrising UA. A 12-year follow-up in sporadic inclusion body myositis: an end stage with major disabilities. Brain : a journal of neurology. 2011;134:3167–75. - PubMed
-
- Cortese A, Machado P, Morrow J, Dewar L, Hiscock A, Miller A, Brady S, Hilton-Jones D, Parton M, Hanna MG. Longitudinal observational study of sporadic inclusion body myositis: implications for clinical trials. Neuromuscular disorders : NMD. 2013;23:404–12. Epub 2013/03/16. - PubMed
-
- Askanas V, Engel WK, Nogalska A. Pathogenic considerations in sporadic inclusion-body myositis, a degenerative muscle disease associated with aging and abnormalities of myoproteostasis. Journal of neuropathology and experimental neurology. 2012;71:680–93. Epub 2012/07/19. - PubMed
-
- Vattemi G, Nogalska A, King Engel W, D’Agostino C, Checler F, Askanas V. Amyloid-beta42 is preferentially accumulated in muscle fibers of patients with sporadic inclusion-body myositis. Acta neuropathologica. 2009;117:569–74. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

