Puberty-specific promotion of mammary tumorigenesis by a high animal fat diet
- PMID: 26526858
- PMCID: PMC4630903
- DOI: 10.1186/s13058-015-0646-4
Puberty-specific promotion of mammary tumorigenesis by a high animal fat diet
Abstract
Introduction: Increased animal fat consumption is associated with increased premenopausal breast cancer risk in normal weight, but not overweight, women. This agrees with our previous findings in obesity-resistant BALB/c mice, in which exposure to a high saturated animal fat diet (HFD) from peripuberty through adulthood promoted mammary tumorigenesis. Epidemiologic and animal studies support the importance of puberty as a life stage when diet and environmental exposures affect adult breast cancer risk. In this study, we identified the effects of peripubertal exposure to HFD and investigated its mechanism of enhancing tumorigenesis.
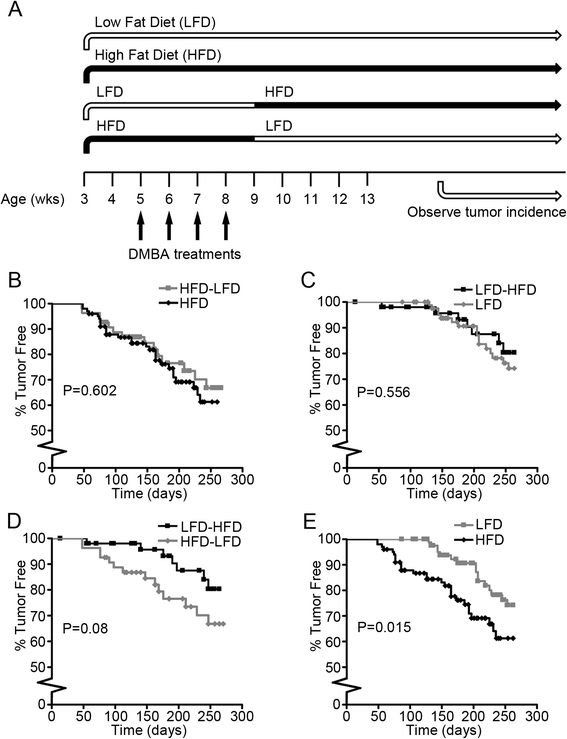
Methods: Three-week-old BALB/c mice fed a low-fat diet (LFD) or HFD were subjected to 7,12-dimethylbenz[a]anthracene (DMBA)-induced carcinogenesis. At 9 weeks of age, half the mice on LFD were switched to HFD (LFD-HFD group) and half the mice on HFD were switched to LFD (HFD-LFD group). Tumor gene expression was evaluated in association with diet and tumor latency.
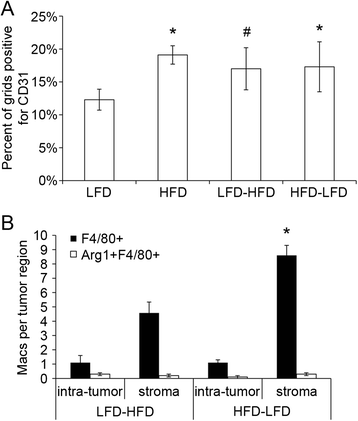
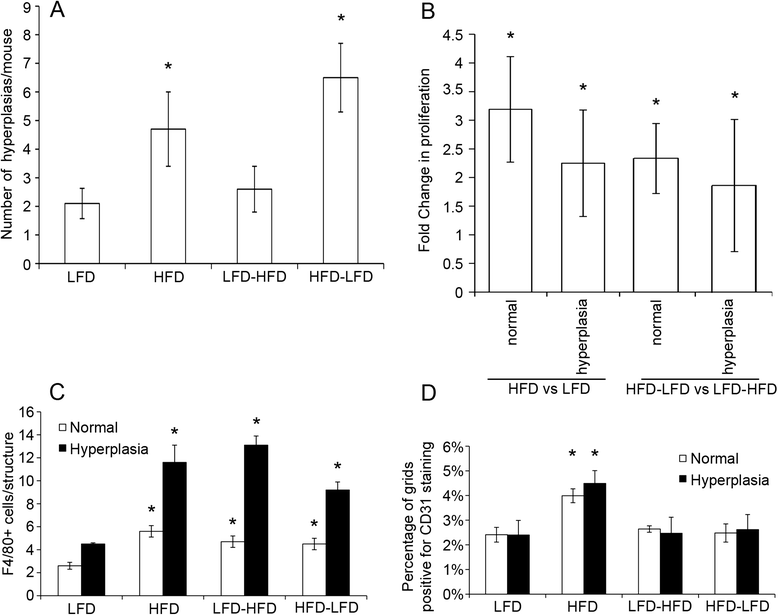
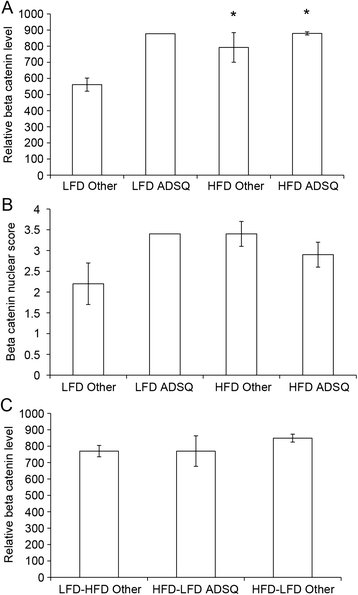
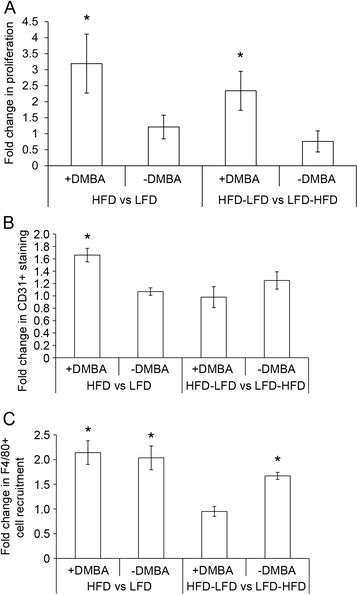
Results: The peripubertal HFD reduced the latency of DMBA-induced mammary tumors and was associated with tumor characteristics similar to those in mice fed a continuous HFD. Notably, short-latency tumors in both groups shared gene expression characteristics and were more likely to have adenosquamous histology. Both HFD-LFD and continuous HFD tumors showed similar gene expression patterns and early latency. Adult switch from HFD to LFD did not reverse peripubertal HFD tumor promotion. Increased proliferation, hyperplasia, and macrophages were present in mammary glands before tumor development, implicating these as possible effectors of tumor promotion. Despite a significant interaction between pubertal diet and carcinogens in tumor promotion, peripubertal HFD by itself produced persistent macrophage recruitment to mammary glands.
Conclusions: In obesity-resistant mice, peripubertal HFD is sufficient to irreversibly promote carcinogen-induced tumorigenesis. Increased macrophage recruitment is likely a contributing factor. These results underscore the importance of early life exposures to increased adult cancer risk and are consistent with findings that an HFD in normal weight premenopausal women leads to increased breast cancer risk. Notably, short-latency tumors occurring after peripubertal HFD had characteristics similar to human basal-like breast cancers that predominantly develop in younger women.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

