Application effect of extensively hydrolyzed milk protein formula and follow-up in preterm children with a gestational age of less than 34 weeks: study protocol for a randomized controlled trial
- PMID: 26537897
- PMCID: PMC4632355
- DOI: 10.1186/s13063-015-1030-5
Application effect of extensively hydrolyzed milk protein formula and follow-up in preterm children with a gestational age of less than 34 weeks: study protocol for a randomized controlled trial
Abstract
Background: The average incidence of preterm birth in the world is up to 11.1 %, and deaths of preterm children account for more than 50 % of neonatal deaths. Gastrointestinal function of preterm children with a gestational age less than 34 weeks is immaturely developed. For preterm children who can only be fed with formula due to their mothers' sickness, choosing a suitable formula can not only meet the high nutritional needs of preterm children, but also solve their low gastrointestinal tolerability, and is thus very important.
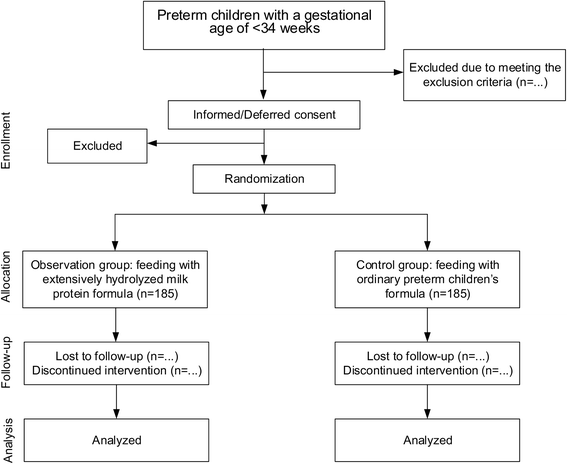
Methods/design: The study is a prospective, randomized, single-blind and controlled clinical trial. Preterm children with a gestational age less than 34 weeks meeting the inclusion criteria who cannot be breastfed will be included. To demonstrate the application effect of extensively hydrolyzed milk protein formula on the target population, preterm children will be randomized into two groups, 185 subjects in each group. The observation group will be fed with extensively hydrolyzed milk protein (100 % whey protein) formula, while the control group will be fed with preterm children's formula until the children are discharged from the neonatal intensive care unit (NICU). All the formula involved in this study will be from Dumex. After discharge, both groups will be uniformly fed with formula for 0 to 6-month-old infants. For statistical analysis, a chi-square test and Student's t test will be applied using SAS 9.4.
Discussion: This will be the first randomized controlled clinical study with long-term observation of the growth and development of preterm children during the NICU stay and at 3-month follow-up after discharge from the NICU. Results from this study will be used to determine whether the extensively hydrolyzed formula is more suitable for the low gastrointestinal tolerability of preterm children, and also whether feeding preterm children who are fed with such formula during the NICU stay with ordinary infant formula after discharge from the NICU would affect the normal growth and development of preterm children in the early stage of their lives.
Trial registration: This study was registered with the Chinese Clinical Trial Registry ( http://www.chictr.org.cn/ ) with number ChiCTR-IOR-14005696 , on December 22, 2014.
Figures
References
-
- World Health Organization Recommended definitions, terminology and format for statistical tables related to the perinatal period and use of a new certificate for cause of perinatal deaths. Modifications recommended by FIGO as amended October 14, 1976. Acta Obstet Gynecol Scand. 1977;56:247–53. - PubMed
-
- Howson C, Kinney M, Lawn J. Born too soon: the global action report on preterm birth. Geneva: World Health Organization; 2012.
-
- Blencowe H, Cousens S, Oestergaard MZ, Chou D, Moller A, Narwal R, et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: a systematic analysis and implications. Lancet. 2012;379:2162–72. doi: 10.1016/S0140-6736(12)60820-4. - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical