Primary cilia are critical for Sonic hedgehog-mediated dopaminergic neurogenesis in the embryonic midbrain
- PMID: 26542012
- PMCID: PMC5873949
- DOI: 10.1016/j.ydbio.2015.10.033
Primary cilia are critical for Sonic hedgehog-mediated dopaminergic neurogenesis in the embryonic midbrain
Abstract
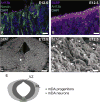
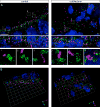
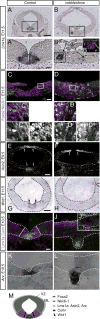
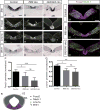
Midbrain dopaminergic (mDA) neurons modulate various motor and cognitive functions, and their dysfunction or degeneration has been implicated in several psychiatric diseases. Both Sonic Hedgehog (Shh) and Wnt signaling pathways have been shown to be essential for normal development of mDA neurons. Primary cilia are critical for the development of a number of structures in the brain by serving as a hub for essential developmental signaling cascades, but their role in the generation of mDA neurons has not been examined. We analyzed mutant mouse lines deficient in the intraflagellar transport protein IFT88, which is critical for primary cilia function. Conditional inactivation of Ift88 in the midbrain after E9.0 results in progressive loss of primary cilia, a decreased size of the mDA progenitor domain, and a reduction in mDA neurons. We identified Shh signaling as the primary cause of these defects, since conditional inactivation of the Shh signaling pathway after E9.0, through genetic ablation of Gli2 and Gli3 in the midbrain, results in a phenotype basically identical to the one seen in Ift88 conditional mutants. Moreover, the expansion of the mDA progenitor domain observed when Shh signaling is constitutively activated does not occur in absence of Ift88. In contrast, clusters of Shh-responding progenitors are maintained in the ventral midbrain of the hypomorphic Ift88 mouse mutant, cobblestone. Despite the residual Shh signaling, the integrity of the mDA progenitor domain is severely disturbed, and consequently very few mDA neurons are generated in cobblestone mutants. Our results identify for the first time a crucial role of primary cilia in the induction of mDA progenitors, define a narrow time window in which Shh-mediated signaling is dependent upon normal primary cilia function for this purpose, and suggest that later Wnt signaling-dependent events act independently of primary cilia.
Keywords: Dopaminergic neurons; Ift88; Intraflagellar transport; Midbrain; Primary cilia; Shh.
Copyright © 2015 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare no competing financial interests or other conflicts of interest.
Figures







Similar articles
-
Definition of a critical spatiotemporal window within which primary cilia control midbrain dopaminergic neurogenesis.Neurogenesis (Austin). 2016 Oct 20;3(1):e1248206. doi: 10.1080/23262133.2016.1248206. eCollection 2016. Neurogenesis (Austin). 2016. PMID: 28090543 Free PMC article.
-
Intraflagellar transport protein 122 antagonizes Sonic Hedgehog signaling and controls ciliary localization of pathway components.Proc Natl Acad Sci U S A. 2011 Jan 25;108(4):1456-61. doi: 10.1073/pnas.1011410108. Epub 2011 Jan 5. Proc Natl Acad Sci U S A. 2011. PMID: 21209331 Free PMC article.
-
Temporal and spatial requirements of Smoothened in ventral midbrain neuronal development.Neural Dev. 2013 Apr 26;8:8. doi: 10.1186/1749-8104-8-8. Neural Dev. 2013. PMID: 23618354 Free PMC article.
-
Wnt/β-catenin signaling in midbrain dopaminergic neuron specification and neurogenesis.J Mol Cell Biol. 2014 Feb;6(1):27-33. doi: 10.1093/jmcb/mjt043. Epub 2013 Nov 28. J Mol Cell Biol. 2014. PMID: 24287202 Review.
-
Regulatory mechanisms governing GLI proteins in hedgehog signaling.Anat Sci Int. 2025 Mar;100(2):143-154. doi: 10.1007/s12565-024-00814-1. Epub 2024 Nov 22. Anat Sci Int. 2025. PMID: 39576500 Review.
Cited by
-
Multi-omic analysis of the ciliogenic transcription factor RFX3 reveals a role in promoting activity-dependent responses via enhancing CREB binding in human neurons.bioRxiv [Preprint]. 2025 Mar 1:2025.02.27.640588. doi: 10.1101/2025.02.27.640588. bioRxiv. 2025. PMID: 40060598 Free PMC article. Preprint.
-
Fibroblast Primary Cilia Are Required for Cardiac Fibrosis.Circulation. 2019 May 14;139(20):2342-2357. doi: 10.1161/CIRCULATIONAHA.117.028752. Circulation. 2019. PMID: 30818997 Free PMC article.
-
Ciliary and Non-Ciliary Roles of IFT88 in Development and Diseases.Int J Mol Sci. 2025 Feb 27;26(5):2110. doi: 10.3390/ijms26052110. Int J Mol Sci. 2025. PMID: 40076734 Free PMC article. Review.
-
Recent advances in the understanding of cilia mechanisms and their applications as therapeutic targets.Front Mol Biosci. 2023 Sep 14;10:1232188. doi: 10.3389/fmolb.2023.1232188. eCollection 2023. Front Mol Biosci. 2023. PMID: 37780208 Free PMC article. Review.
-
Hedging against Neuropathic Pain: Role of Hedgehog Signaling in Pathological Nerve Healing.Int J Mol Sci. 2020 Nov 30;21(23):9115. doi: 10.3390/ijms21239115. Int J Mol Sci. 2020. PMID: 33266112 Free PMC article. Review.
References
-
- Abdelhamed ZA, Wheway G, Szymanska K, Natarajan S, Toomes C, Inglehearn C, Johnson CA. Variable expressivity of ciliopathy neurological phenotypes that encompass Meckel-Gruber syndrome and Joubert syndrome is caused by complex de-regulated ciliogenesis, Shh and Wnt signalling defects. Hum. Mol. Genet. 2013;22:1358–1372. http://dx.doi.org/10.1093/hmg/dds546. - DOI - PMC - PubMed
-
- Albin RL, Young AB, Penney JB. The functional-anatomy of basal ganglia disorders. Trends Neurosci. 1989;12:366–375. - PubMed
-
- Andersson E, Tryggvason U, Deng Q, Friling S, Alekseenko Z, Robert B, Perlmann T, Ericson J. Identification of intrinsic determinants of midbrain dopamine neurons. Cell. 2006;124:393–405. http://dx.doi.org/10.1016/j.cell.2005.10.037. - DOI - PubMed
-
- Andersson ER, Saltó C, Villaescusa JC, Cajanek L, Yang S, Bryjova L, Nagy II, Vainio SJ, Ramirez C, Bryja V. Wnt5a cooperates with canonical Wnts to generate midbrain dopaminergic neurons in vivo and in stem cells. Proc. Natl. Acad. Sci. USA. 2013;110:E602–E610. http://dx.doi.org/10.1073/pnas.1208524110/-/DCSupplemental. - DOI - PMC - PubMed
-
- Baala L, Romano S, Khaddour R, Saunier S, Smith UM, Audollent S, Ozilou C, Faivre L, Laurent N, Foliguet B, Munnich A, Lyonnet S, Salomon R, EnchaRazavi F, Gubler M-C, Boddaert N, de Lonlay P, Johnson CA, Vekemans M, Antignac C, Attié-Bitach T. The Meckel-Gruber syndrome gene, MKS3, is mutated in Joubert syndrome. Am. J. Hum. Genet. 2006;80:186–194. http://dx.doi.org/10.1086/510499. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

