Insights into the Molecular Mechanism of Polymerization and Nucleoside Reverse Transcriptase Inhibitor Incorporation by Human PrimPol
- PMID: 26552983
- PMCID: PMC4704227
- DOI: 10.1128/AAC.02270-15
Insights into the Molecular Mechanism of Polymerization and Nucleoside Reverse Transcriptase Inhibitor Incorporation by Human PrimPol
Abstract
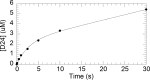
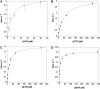
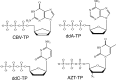
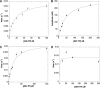
Human PrimPol is a newly identified DNA and RNA primase-polymerase of the archaeo-eukaryotic primase (AEP) superfamily and only the second known polymerase in the mitochondria. Mechanistic studies have shown that interactions of the primary mitochondrial DNA polymerase γ (mtDNA Pol γ) with nucleoside reverse transcriptase inhibitors (NRTIs), key components in treating HIV infection, are a major source of NRTI-associated toxicity. Understanding the interactions of host polymerases with antiviral and anticancer nucleoside analog therapies is critical for preventing life-threatening adverse events, particularly in AIDS patients who undergo lifelong treatment. Since PrimPol has only recently been discovered, the molecular mechanism of polymerization and incorporation of natural nucleotide and NRTI substrates, crucial for assessing the potential for PrimPol-mediated NRTI-associated toxicity, has not been explored. We report for the first time a transient-kinetic analysis of polymerization for each nucleotide and NRTI substrate as catalyzed by PrimPol. These studies reveal that nucleotide selectivity limits chemical catalysis while the release of the elongated DNA product is the overall rate-limiting step. Remarkably, PrimPol incorporates four of the eight FDA-approved antiviral NRTIs with a kinetic profile distinct from that of mtDNA Pol γ that may manifest in toxicity.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures


Similar articles
-
Mitochondrial DNA replication: a PrimPol perspective.Biochem Soc Trans. 2017 Apr 15;45(2):513-529. doi: 10.1042/BST20160162. Biochem Soc Trans. 2017. PMID: 28408491 Free PMC article. Review.
-
Identifying the role of PrimPol in TDF-induced toxicity and implications of its loss of function mutation in an HIV+ patient.Sci Rep. 2020 Jun 9;10(1):9343. doi: 10.1038/s41598-020-66153-z. Sci Rep. 2020. PMID: 32518272 Free PMC article.
-
Divalent Cations Alter the Rate-Limiting Step of PrimPol-Catalyzed DNA Elongation.J Mol Biol. 2019 Feb 15;431(4):673-686. doi: 10.1016/j.jmb.2019.01.002. Epub 2019 Jan 8. J Mol Biol. 2019. PMID: 30633872 Free PMC article.
-
Optimization of the expression, purification and polymerase activity reaction conditions of recombinant human PrimPol.PLoS One. 2017 Sep 13;12(9):e0184489. doi: 10.1371/journal.pone.0184489. eCollection 2017. PLoS One. 2017. PMID: 28902865 Free PMC article.
-
Mechanism of DNA primer synthesis by human PrimPol.Enzymes. 2019;45:289-310. doi: 10.1016/bs.enz.2019.06.003. Epub 2019 Jul 13. Enzymes. 2019. PMID: 31627881 Review.
Cited by
-
3'dNTP Binding Is Modulated during Primer Synthesis and Translesion by Human PrimPol.Int J Mol Sci. 2023 Dec 19;25(1):51. doi: 10.3390/ijms25010051. Int J Mol Sci. 2023. PMID: 38203225 Free PMC article.
-
Human PrimPol Discrimination against Dideoxynucleotides during Primer Synthesis.Genes (Basel). 2021 Sep 24;12(10):1487. doi: 10.3390/genes12101487. Genes (Basel). 2021. PMID: 34680882 Free PMC article.
-
DNA polymerases in the mitochondria: A critical review of the evidence.Front Biosci (Landmark Ed). 2017 Jan 1;22(4):692-709. doi: 10.2741/4510. Front Biosci (Landmark Ed). 2017. PMID: 27814640 Free PMC article. Review.
-
Mitochondrial DNA replication: a PrimPol perspective.Biochem Soc Trans. 2017 Apr 15;45(2):513-529. doi: 10.1042/BST20160162. Biochem Soc Trans. 2017. PMID: 28408491 Free PMC article. Review.
-
Mechanisms of direct replication restart at stressed replisomes.DNA Repair (Amst). 2020 Nov;95:102947. doi: 10.1016/j.dnarep.2020.102947. Epub 2020 Aug 16. DNA Repair (Amst). 2020. PMID: 32853827 Free PMC article. Review. No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

