Modulation of expression of heat shock proteins and apoptosis by Flueggea leucopyrus (Willd) decoction in three breast cancer phenotypes
- PMID: 26553005
- PMCID: PMC4640413
- DOI: 10.1186/s12906-015-0927-6
Modulation of expression of heat shock proteins and apoptosis by Flueggea leucopyrus (Willd) decoction in three breast cancer phenotypes
Abstract
Background: During the past few years, there has been an increasing interest among the Traditional and Folk medical practitioners of Sri Lanka in the use of a decoction prepared from Flueggea leucopyrus (Willd.) for treating various cancers including breast cancer. In the present study, the cytotoxicity of this decoction and its effects on Heat Shock Protein (HSP) expression and apoptosis were compared in three breast cancer phenotypes, to scientifically evaluate if a decoction prepared from F. leucopyrus (Willd.) is useful for the treatment of breast cancer.
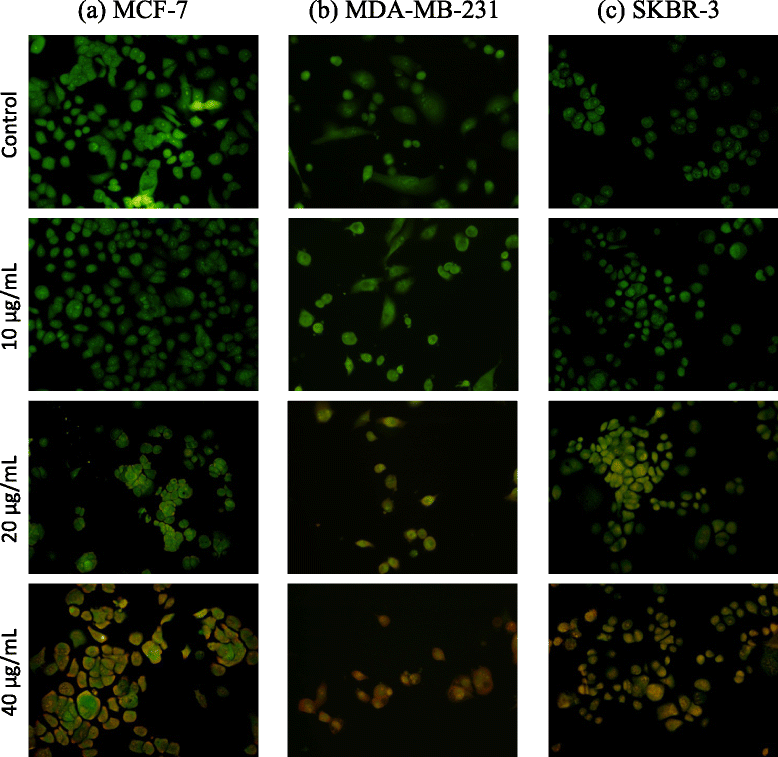
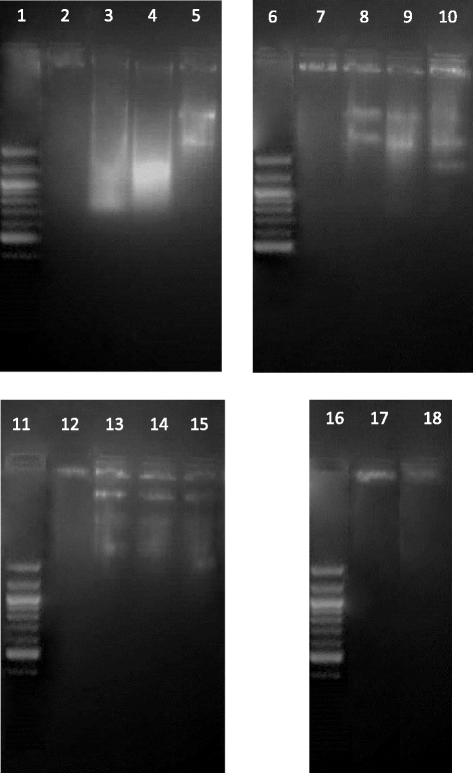
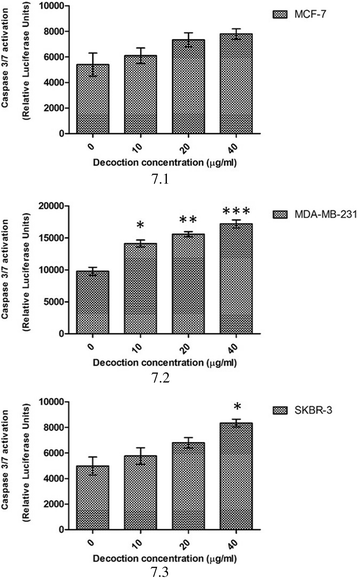
Methods: Cytotoxic potential of the F. leucopyrus decoction was determined by evaluating its effects in MCF-7, MDA-MB-231 and SKBR-3 breast cancer cell lines, and MCF-10A (non-cancerous) breast cell line, by use of the Sulphorhodamine (SRB) assay. The effect of the decoction on HSP gene expression in the above cells was evaluated by (a) Real time reverse transcription PCR (RT-PCR) and (b) Immunofluorescence analysis of HSP protein expression. Effects of the decoction on apoptosis were evaluated by (a) fluorescent microscopic examination of apoptosis related morphological changes and (b) DNA fragmentation (c) Caspase 3/7 assay.
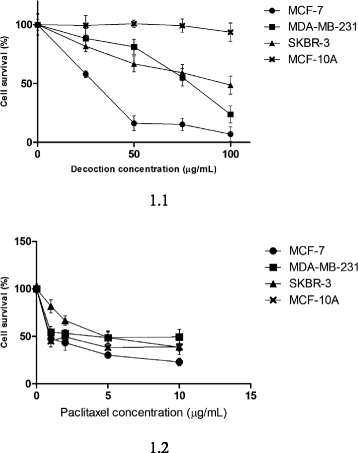
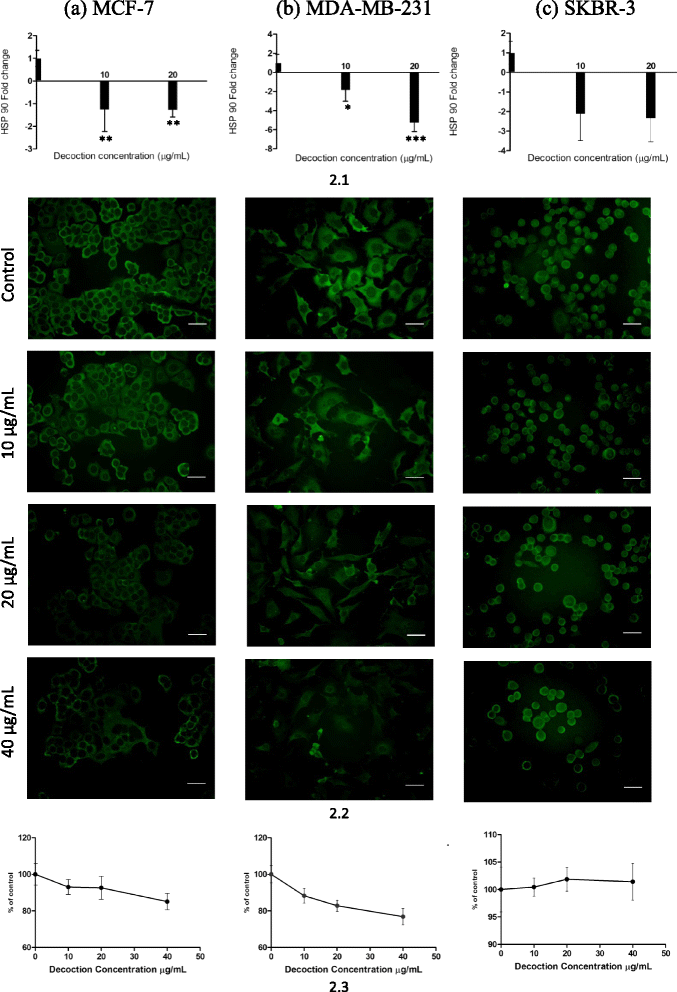
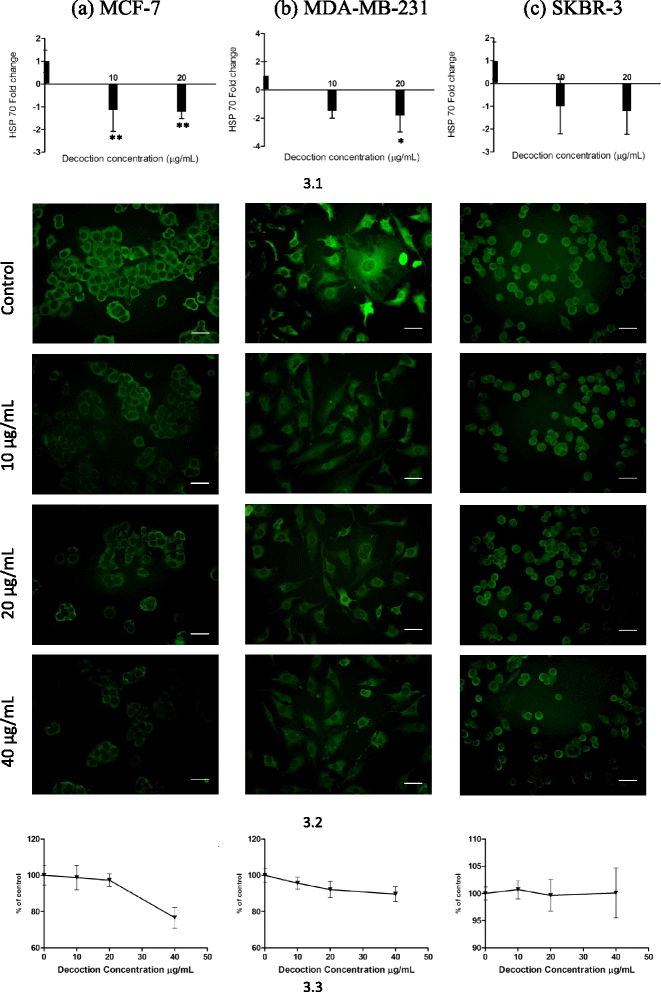
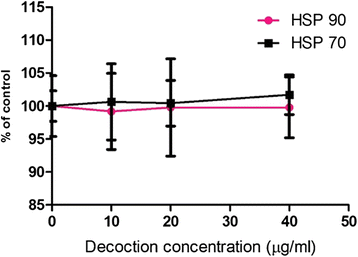
Results: F. leucopyrus decoction can mediate significant cytotoxic effects in all three breast cancer cells phenotypes (IC50 values: 27.89, 99.43, 121.43 μg/mL at 24 h post incubation periods, for MCF-7, MDA-MB-231, SKBR-3 respectively) with little effect in the non-cancerous breast cell line MCF-10A (IC50: 570.4 μg/mL). Significant (*P <0.05) inhibitions of HSP 90 and HSP 70 expression were mediated by the decoction in MCF-7 and MDA-MB-231, with little effect in the SKBR-3 cells. Clear apoptotic morphological changes on Acridine orange/Ethidium bromide staining and DNA fragmentation were observed in all three breast cancer cell lines. Caspase 3/7 were significantly (*P <0.05) activated only in MDA-MB-231 and SKBR-3 cells indicating caspase dependent apoptosis in these cells and caspase independent apoptosis in MCF-7 cells.
Conclusions: Modulation of HSP 90 and HSP 70 expressions is a possible mechanism by which the decoction of F. leucopyrus mediates cytotoxic effects MCF-7 and MDA-MB-231 cells. This effect appears to correlate with enhanced apoptosis in these cells. In SKBR-3 cells, mechanisms other than HSP inhibition may be utilized to a greater extent by the decoction to mediate the observed cytotoxic effects. Overall findings suggest that the decoction has the potential to be exploited further for effective treatment of breast cancer.
Figures
References
-
- Roberts CG, Gurusik E, Biden TJ, Sutherland RL, Butt AJ. Synergistic cytotoxicity between tamoxifen and the plant toxin persin in human breast cancer cells is dependent on Bim expression and mediated by modulation of ceramide metabolism. Mol Cancer Ther. 2007;6:2777–2785. doi: 10.1158/1535-7163.MCT-07-0374. - DOI - PubMed
-
- Altenberg JD, Bieberich AA, Terry C, Harvey KA, VanHom JF, Xu Z, Davisson VJ, Siddiqui RA. A synergestic antiproliferation effect of curcumin and docosahexannoic acid in SKBR-3 breast cancer cells: unique signalling not explained by the effects of either compound alone. BMC Cancer. 2011;11:149–165. doi: 10.1186/1471-2407-11-149. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

