Inactivation of Peroxiredoxin 6 by the Pla Protease of Yersinia pestis
- PMID: 26553463
- PMCID: PMC4693992
- DOI: 10.1128/IAI.01168-15
Inactivation of Peroxiredoxin 6 by the Pla Protease of Yersinia pestis
Abstract
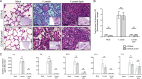
Pneumonic plague represents the most severe form of disease caused by Yersinia pestis due to its ease of transmission, rapid progression, and high mortality rate. The Y. pestis outer membrane Pla protease is essential for the development of pneumonic plague; however, the complete repertoire of substrates cleaved by Pla in the lungs is not known. In this study, we describe a proteomic screen to identify host proteins contained within the bronchoalveolar lavage fluid of mice that are cleaved and/or processed by Y. pestis in a Pla-dependent manner. We identified peroxiredoxin 6 (Prdx6), a host factor that contributes to pulmonary surfactant metabolism and lung defense against oxidative stress, as a previously unknown substrate of Pla. Pla cleaves Prdx6 at three distinct sites, and these cleavages disrupt both the peroxidase and phospholipase A2 activities of Prdx6. In addition, we found that infection with wild-type Y. pestis reduces the abundance of extracellular Prdx6 in the lungs compared to that after infection with Δpla Y. pestis, suggesting that Pla cleaves Prdx6 in the pulmonary compartment. However, following infection with either wild-type or Δpla Y. pestis, Prdx6-deficient mice exhibit no differences in bacterial burden, host immune response, or lung damage from wild-type mice. Thus, while Pla is able to disrupt Prdx6 function in vitro and reduce Prdx6 levels in vivo, the cleavage of Prdx6 has little detectable impact on the progression or outcome of pneumonic plague.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures

Similar articles
-
Proteolysis of plasminogen activator inhibitor-1 by Yersinia pestis remodulates the host environment to promote virulence.J Thromb Haemost. 2016 Sep;14(9):1833-43. doi: 10.1111/jth.13408. Epub 2016 Aug 19. J Thromb Haemost. 2016. PMID: 27377187 Free PMC article.
-
Yersinia pestis Plasminogen Activator.Biomolecules. 2020 Nov 14;10(11):1554. doi: 10.3390/biom10111554. Biomolecules. 2020. PMID: 33202679 Free PMC article. Review.
-
Impact of the Pla protease substrate α2-antiplasmin on the progression of primary pneumonic plague.Infect Immun. 2015 Dec;83(12):4837-47. doi: 10.1128/IAI.01086-15. Epub 2015 Oct 5. Infect Immun. 2015. PMID: 26438794 Free PMC article.
-
Depletion of Glucose Activates Catabolite Repression during Pneumonic Plague.J Bacteriol. 2018 May 9;200(11):e00737-17. doi: 10.1128/JB.00737-17. Print 2018 Jun 1. J Bacteriol. 2018. PMID: 29555700 Free PMC article.
-
Fibrinolytic and coagulative activities of Yersinia pestis.Front Cell Infect Microbiol. 2013 Jul 26;3:35. doi: 10.3389/fcimb.2013.00035. eCollection 2013. Front Cell Infect Microbiol. 2013. PMID: 23898467 Free PMC article. Review.
Cited by
-
Unveiling the dance of evolution: Pla-mediated cleavage of Ymt modulates the virulence dynamics of Yersinia pestis.mBio. 2024 Aug 14;15(8):e0107524. doi: 10.1128/mbio.01075-24. Epub 2024 Jul 3. mBio. 2024. PMID: 38958447 Free PMC article.
-
A Dual Role for the Plasminogen Activator Protease During the Preinflammatory Phase of Primary Pneumonic Plague.J Infect Dis. 2020 Jul 6;222(3):407-416. doi: 10.1093/infdis/jiaa094. J Infect Dis. 2020. PMID: 32128567 Free PMC article.
-
Proteolysis of plasminogen activator inhibitor-1 by Yersinia pestis remodulates the host environment to promote virulence.J Thromb Haemost. 2016 Sep;14(9):1833-43. doi: 10.1111/jth.13408. Epub 2016 Aug 19. J Thromb Haemost. 2016. PMID: 27377187 Free PMC article.
-
Yersinia pestis Plasminogen Activator.Biomolecules. 2020 Nov 14;10(11):1554. doi: 10.3390/biom10111554. Biomolecules. 2020. PMID: 33202679 Free PMC article. Review.
-
Two Isoforms of Yersinia pestis Plasminogen Activator Pla: Intraspecies Distribution, Intrinsic Disorder Propensity, and Contribution to Virulence.PLoS One. 2016 Dec 9;11(12):e0168089. doi: 10.1371/journal.pone.0168089. eCollection 2016. PLoS One. 2016. PMID: 27936190 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

