Evolutionarily conserved mechanisms for the selection and maintenance of behavioural activity
- PMID: 26554043
- PMCID: PMC4650127
- DOI: 10.1098/rstb.2015.0053
Evolutionarily conserved mechanisms for the selection and maintenance of behavioural activity
Abstract
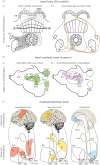
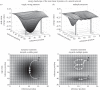
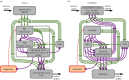
Survival and reproduction entail the selection of adaptive behavioural repertoires. This selection manifests as phylogenetically acquired activities that depend on evolved nervous system circuitries. Lorenz and Tinbergen already postulated that heritable behaviours and their reliable performance are specified by genetically determined programs. Here we compare the functional anatomy of the insect central complex and vertebrate basal ganglia to illustrate their role in mediating selection and maintenance of adaptive behaviours. Comparative analyses reveal that central complex and basal ganglia circuitries share comparable lineage relationships within clusters of functionally integrated neurons. These clusters are specified by genetic mechanisms that link birth time and order to their neuronal identities and functions. Their subsequent connections and associated functions are characterized by similar mechanisms that implement dimensionality reduction and transition through attractor states, whereby spatially organized parallel-projecting loops integrate and convey sensorimotor representations that select and maintain behavioural activity. In both taxa, these neural systems are modulated by dopamine signalling that also mediates memory-like processes. The multiplicity of similarities between central complex and basal ganglia suggests evolutionarily conserved computational mechanisms for action selection. We speculate that these may have originated from ancestral ground pattern circuitries present in the brain of the last common ancestor of insects and vertebrates.
Keywords: action selection; attractor state; basal ganglia; brain evolution; central complex; sensorimotor representation.
© 2015 The Authors.
Figures
Similar articles
-
Deep homology of arthropod central complex and vertebrate basal ganglia.Science. 2013 Apr 12;340(6129):157-61. doi: 10.1126/science.1231828. Science. 2013. PMID: 23580521 Review.
-
Evolutionary conservation of the basal ganglia as a common vertebrate mechanism for action selection.Curr Biol. 2011 Jul 12;21(13):1081-91. doi: 10.1016/j.cub.2011.05.001. Epub 2011 Jun 23. Curr Biol. 2011. PMID: 21700460
-
Homology versus convergence in resolving transphyletic correspondences of brain organization.Brain Behav Evol. 2013;82(4):215-9. doi: 10.1159/000356102. Epub 2013 Nov 26. Brain Behav Evol. 2013. PMID: 24296550
-
The Sea Slug, Pleurobranchaea californica: A Signpost Species in the Evolution of Complex Nervous Systems and Behavior.Integr Comp Biol. 2015 Dec;55(6):1058-69. doi: 10.1093/icb/icv081. Epub 2015 Jul 10. Integr Comp Biol. 2015. PMID: 26163678 Free PMC article. Review.
-
Structural, functional and developmental convergence of the insect mushroom bodies with higher brain centers of vertebrates.Brain Behav Evol. 2008;72(1):1-15. doi: 10.1159/000139457. Epub 2008 Jun 17. Brain Behav Evol. 2008. PMID: 18560208
Cited by
-
Neural Organization of the Optic Lobe Changes Steadily from Late Embryonic Stage to Adulthood in Cuttlefish Sepia pharaonis.Front Physiol. 2017 Jul 27;8:538. doi: 10.3389/fphys.2017.00538. eCollection 2017. Front Physiol. 2017. PMID: 28798695 Free PMC article.
-
In vivo expansion of functionally integrated GABAergic interneurons by targeted increase in neural progenitors.EMBO J. 2018 Jul 2;37(13):e98163. doi: 10.15252/embj.201798163. Epub 2018 May 4. EMBO J. 2018. PMID: 29728368 Free PMC article.
-
Ancestral regulatory mechanisms specify conserved midbrain circuitry in arthropods and vertebrates.Proc Natl Acad Sci U S A. 2020 Aug 11;117(32):19544-19555. doi: 10.1073/pnas.1918797117. Epub 2020 Aug 3. Proc Natl Acad Sci U S A. 2020. PMID: 32747566 Free PMC article.
-
Computational models of behavioral addictions: State of the art and future directions.Addict Behav. 2023 May;140:107595. doi: 10.1016/j.addbeh.2022.107595. Epub 2022 Dec 22. Addict Behav. 2023. PMID: 36621045 Free PMC article. Review.
-
In silico Interrogation of Insect Central Complex Suggests Computational Roles for the Ellipsoid Body in Spatial Navigation.Front Behav Neurosci. 2017 Aug 3;11:142. doi: 10.3389/fnbeh.2017.00142. eCollection 2017. Front Behav Neurosci. 2017. PMID: 28824390 Free PMC article.
References
-
- Stirling P, Laughlin S. 2015. Principles of neural design. London, UK: MIT Press.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

