Characterization of mesenchymal stem cells derived from the equine synovial fluid and membrane
- PMID: 26555093
- PMCID: PMC4640348
- DOI: 10.1186/s12917-015-0531-5
Characterization of mesenchymal stem cells derived from the equine synovial fluid and membrane
Abstract
Background: Isolation of mesenchymal stem cells (MSCs) in equines, has been reported for different tissues including bone marrow, adipose, umbilical cord, peripheral blood, and yolk sac. In regard to the MSCs derived from synovial fluid (SF) or membrane (SM), there is data available for humans, dogs, pigs, goats and horses. Especially in equines, these cells have being considered promising candidates for articular regeneration. Herein, we established and characterized MSCs obtained from equine SF and SM. Samples were obtained during arthroscopy and cultured using MEM (Minimum Essential Medium). MSCs were characterized by morphology and expression of specific markers for stem cells, pluripotency, inflammation, and cell cycle.
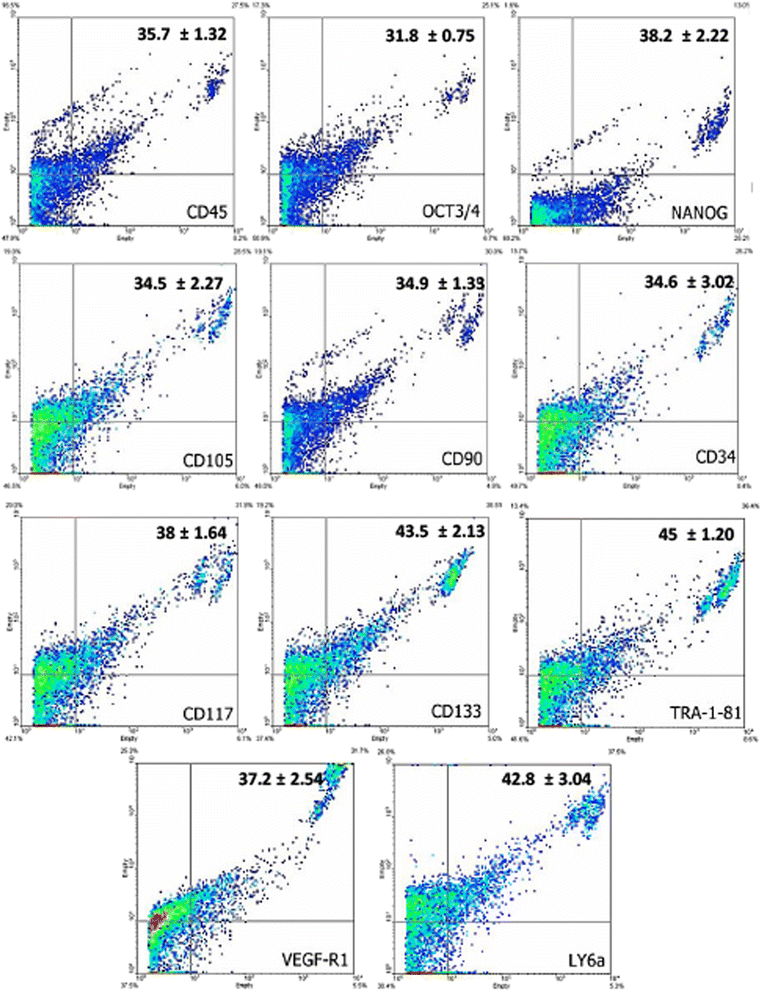
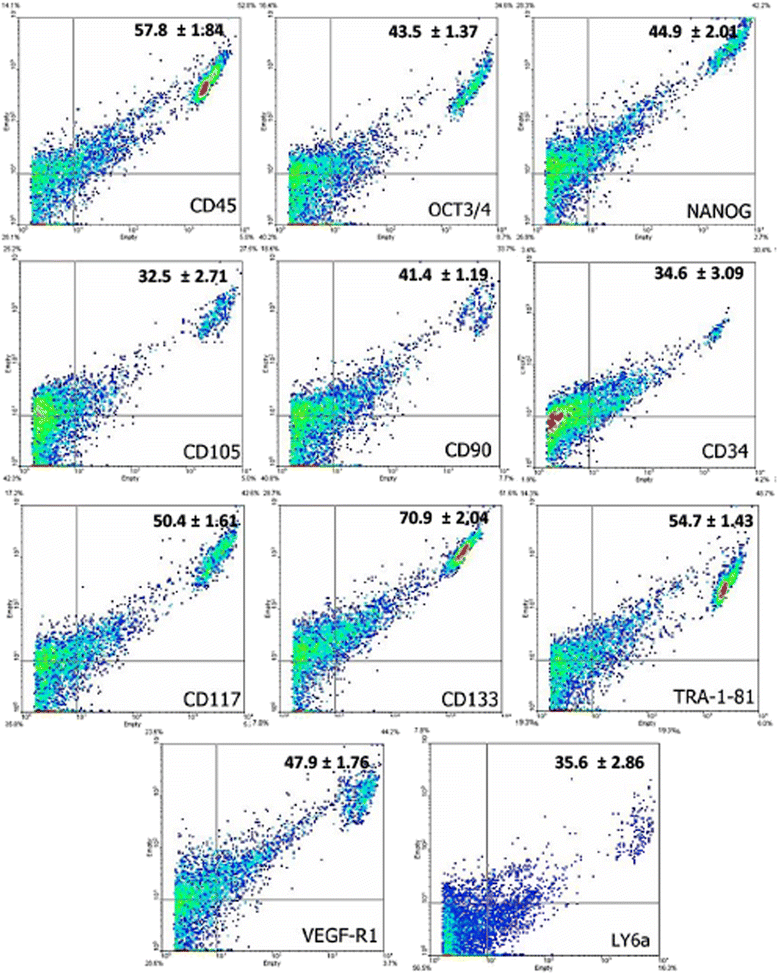
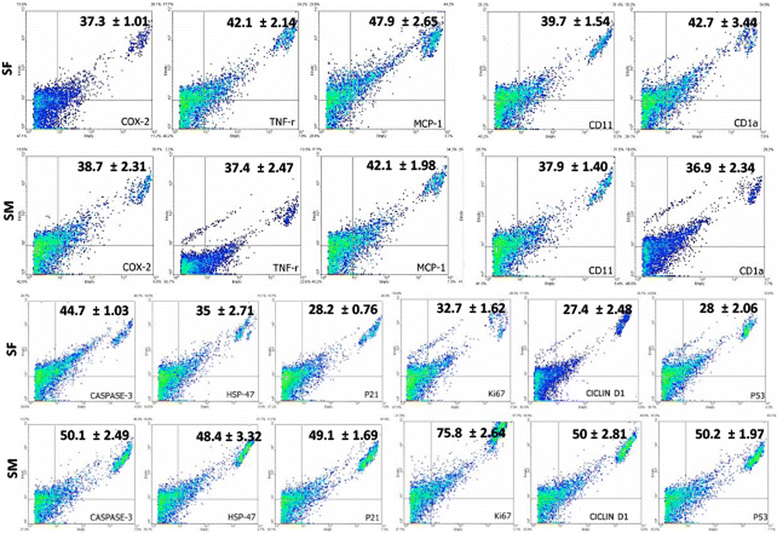
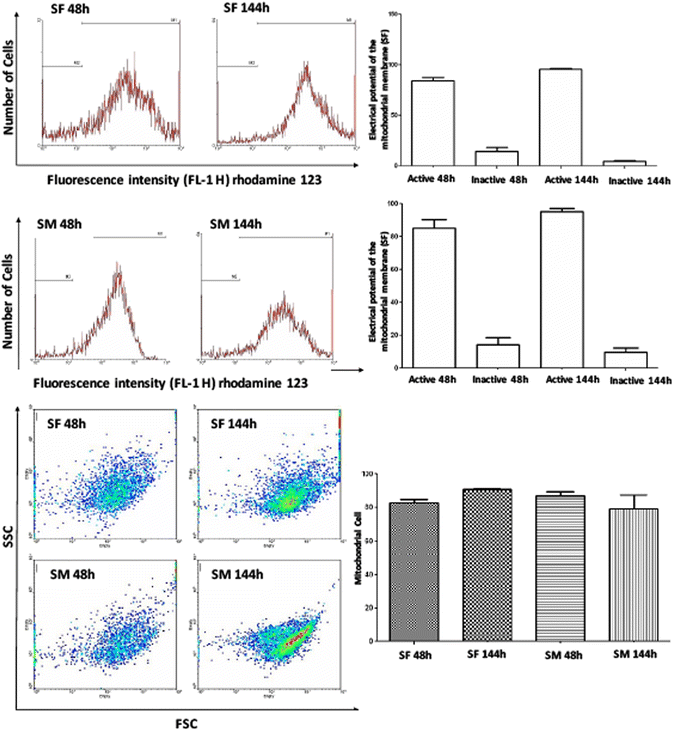
Results: The medium MEM was more effective (97% ± 2) to maintain both cultures. The cultures were composed by adherent cells with fibroblast-like shape, which had a growth pattern represented by a sigmoidal curve. After the expansion, the cells were analyzed by flow cytometry for stem cells, inflammatory, and cell cycle markers, and both lineages showed significant expression of CD45, Oct3/4, Nanog, CD105, CD90, CD34, CD117, CD133, TRA-1-81, VEGF, and LY6a. In contrast, there were differences in the cell cycle phases between the lineages, which was not observed in relation to the mitochondrial electrical potential.
Conclusion: Given the large impact that joint pathology has on the athletic performance horses, our results suggested that the SF and SM are promising sources of stem cells with satisfactory characteristics of growth and gene expression that can be used in equine regenerative medicine.
Figures



References
-
- Canto LS, La Corte FD, Brass KE, Ribeiro MD. Frequência de problemas de equilíbrio nos cascos de cavalos crioulos em treinamento. Braz J Vet Res Anim Sci. 2006;43:489–95.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

