Mesenchymal Conversion of Mesothelial Cells Is a Key Event in the Pathophysiology of the Peritoneum during Peritoneal Dialysis
- PMID: 26556413
- PMCID: PMC4590954
- DOI: 10.1155/2014/473134
Mesenchymal Conversion of Mesothelial Cells Is a Key Event in the Pathophysiology of the Peritoneum during Peritoneal Dialysis
Abstract
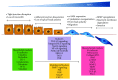
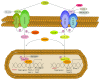
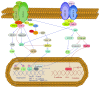
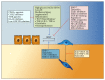
Peritoneal dialysis (PD) is a therapeutic option for the treatment of end-stage renal disease and is based on the use of the peritoneum as a semipermeable membrane for the exchange of toxic solutes and water. Long-term exposure of the peritoneal membrane to hyperosmotic PD fluids causes inflammation, loss of the mesothelial cells monolayer, fibrosis, vasculopathy, and angiogenesis, which may lead to peritoneal functional decline. Peritonitis may further exacerbate the injury of the peritoneal membrane. In parallel with these peritoneal alterations, mesothelial cells undergo an epithelial to mesenchymal transition (EMT), which has been associated with peritoneal deterioration. Factors contributing to the bioincompatibility of classical PD fluids include the high content of glucose/glucose degradation products (GDPs) and their acidic pH. New generation low-GDPs-neutral pH fluids have improved biocompatibility resulting in better preservation of the peritoneum. However, standard glucose-based fluids are still needed, as biocompatible solutions are expensive for many potential users. An alternative approach to preserve the peritoneal membrane, complementary to the efforts to improve fluid biocompatibility, is the use of pharmacological agents protecting the mesothelium. This paper provides a comprehensive review of recent advances that point to the EMT of mesothelial cells as a potential therapeutic target to preserve membrane function.
Figures


References
-
- Chaimovitz C. Peritoneal dialysis. Kidney International. 1994;45(4):1226–1240. - PubMed
-
- Breborowicz A., Oreopoulos D. G. Biocompatibility of peritoneal dialysis solutions. American Journal of Kidney Diseases. 1996;27(5):738–743. - PubMed
-
- Wieczorowska-Tobis K., Polubinska A., Wisniewska J., et al. Multidirectional approach to study peritoneal dialysis fluid biocompatibility in a chronic peritoneal dialysis model in the rat. Nephrology Dialysis Transplantation. 2001;16(3):655–656. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

