A Critical Analysis of Claims and Their Authenticity in Indian Drug Promotional Advertisements
- PMID: 26556557
- PMCID: PMC4590952
- DOI: 10.1155/2015/469147
A Critical Analysis of Claims and Their Authenticity in Indian Drug Promotional Advertisements
Abstract
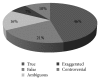
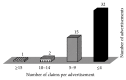
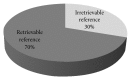
Introduction. Drug promotional advertisements (DPAs) form a major marketing technique of pharmaceutical companies for promoting their products and disseminating ambiguous drug information which can affect prescribing pattern of physicians. Drug information includes product characteristics, various marketing claims with references in support to increase its credibility and authenticity. Material and Methods. An observational study was carried out on fifty printed drug advertisement brochures which were collected from different OPDs of Guru Nanak Dev Hospital attached to Government Medical College, Amritsar, India. These advertisements were analyzed and claims were categorized into true, false, exaggerated, vague, and controversial on criteria as reported by Rohraa et al. (2006). References of DPAs in support of the claims were critically analyzed for their retrievability from web and validity pertaining to claims. Results. Out of 209 claims from 50 advertisements, only 46% were found to be true, 21% false, 16% vague, 7% exaggerated, and 10% controversial in nature. Out of 160 references given in support of claims, 49 (30%) of references were irretrievable. Out of 111 (70%) retrievable references, 92 (83%) references were found valid. Conclusion. Drug information provided in the DPAs was biased, incomplete, unauthentic, and unreliable with references exhibiting questionable credibility.
Figures
Similar articles
-
Evaluation of rationality of promotional drug literature using World Health Organization guidelines.Indian J Pharmacol. 2010 Oct;42(5):267-72. doi: 10.4103/0253-7613.70020. Indian J Pharmacol. 2010. PMID: 21206615 Free PMC article.
-
Critical evaluation of the claims made by pharmaceutical companies in drug promotional material in Pakistan.J Pharm Pharm Sci. 2006;9(1):50-9. J Pharm Pharm Sci. 2006. PMID: 16849008
-
Assessment of Consistency between Claims and References Referred to in Pharmaceutical Advertising Brochures in the Kingdom of Saudi Arabia.Cureus. 2019 Jan 17;11(1):e3907. doi: 10.7759/cureus.3907. Cureus. 2019. PMID: 30931180 Free PMC article.
-
Industry guidelines, laws and regulations ignored: quality of drug advertising in medical journals.Pharmacoepidemiol Drug Saf. 2004 Nov;13(11):789-95. doi: 10.1002/pds.1017. Pharmacoepidemiol Drug Saf. 2004. PMID: 15486957 Review.
-
Natural products and the athlete: facts and folklore.Ann Pharmacother. 1993 May;27(5):607-15. doi: 10.1177/106002809302700516. Ann Pharmacother. 1993. PMID: 8347914 Review.
Cited by
-
Assessment of a Pharmaceutical Advertisement Analysis Module in a Drug Literature Evaluation Course.Am J Pharm Educ. 2017 Aug;81(6):112. doi: 10.5688/ajpe816112. Am J Pharm Educ. 2017. PMID: 28970613 Free PMC article.
-
Critical Appraisal of Drug Promotional Literature in Accordance With WHO Guidelines.Cureus. 2022 Aug 3;14(8):e27644. doi: 10.7759/cureus.27644. eCollection 2022 Aug. Cureus. 2022. PMID: 36072198 Free PMC article.
-
A study of perceptions and exposure of drug promotional literature among clinicians in a teaching hospital.Perspect Clin Res. 2021 Jul-Sep;12(3):140-145. doi: 10.4103/picr.PICR_36_19. Epub 2020 May 7. Perspect Clin Res. 2021. PMID: 34386378 Free PMC article.
References
-
- Thawani V. Drug promotion: can self-regulation work? Indian Journal of Pharmacology. 2002;34(4):227–228.
-
- Sahne B. J., Yegenoglu S., Uner M., Wertheimer A. I. Adherence of drug advertisements to the international marketing codes. Hacettepe University Journal of the Faculty of Pharmacy. 2012;32(1):53–66.
-
- Lal A. Pharmaceutical drug promotion: how it is being practiced in India? The Journal of the Association of Physicians of India. 2001;49:266–273. - PubMed
-
- WHO. Trade, foreign policy, diplomacy and health. http://www.who.int/trade/glossary/story073/en/#main.
LinkOut - more resources
Full Text Sources
Other Literature Sources