A Whole-Body Physiologically Based Pharmacokinetic Model of Gefitinib in Mice and Scale-Up to Humans
- PMID: 26559435
- PMCID: PMC4706283
- DOI: 10.1208/s12248-015-9836-3
A Whole-Body Physiologically Based Pharmacokinetic Model of Gefitinib in Mice and Scale-Up to Humans
Abstract
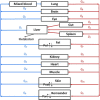
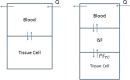
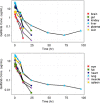
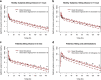
Gefitinib (Iressa) is a selective and potent EGFR tyrosine kinase inhibitor. It received an accelerated FDA approval in 2003 for the treatment of patients with nonsmall cell lung cancer (NSCLC) and represents the first-line therapy for NSCLC with EGFR mutations. In the work presented herein, the disposition of gefitinib was investigated extensively in mouse in both plasma and 11 organs (liver, heart, lung, spleen, gut, brain, skin, fat, eye, kidney, and muscle) after a single IV dose of 20 mg/kg. Gefitinib demonstrated extensive distribution in most tissues, except for the brain, and tissue to plasma partition coefficients (K pt) ranged from 0.71 (brain) to 40.5 (liver). A comprehensive whole-body physiologically based pharmacokinetic (PBPK) model of gefitinib in mice was developed, which adequately captured gefitinib concentration-time profiles in plasma and various tissues. Predicted plasma and tissue AUC values agreed well with the values calculated using the noncompartmental analysis (<25% difference). The PBPK model was further extrapolated to humans after taking into account the interspecies differences in physiological parameters. The simulated concentrations in human plasma were in line with the observed concentrations in healthy volunteers and patients with solid malignant tumors after both IV infusion and oral administration. Considering the extensive tissue distribution of gefitinib, plasma concentration may not be an ideal surrogate marker for gefitinib exposure at the target site or organ of toxicity (such as the skin). Since our whole-body PBPK model can predict gefitinib concentrations not only in plasma but also in various organs, our model may have clinical applications in efficacy and safety assessment of gefitinib.
Keywords: gefitinib; human scale-up; physiologically based pharmacokinetic model; tyrosine kinase inhibitor.
Figures

Similar articles
-
Physiologically Based Pharmacokinetic Modeling to Evaluate the Systemic Exposure of Gefitinib in CYP2D6 Ultrarapid Metabolizers and Extensive Metabolizers.J Clin Pharmacol. 2018 Apr;58(4):485-493. doi: 10.1002/jcph.1036. Epub 2017 Nov 28. J Clin Pharmacol. 2018. PMID: 29193123 Clinical Trial.
-
Single-dose clinical pharmacokinetic studies of gefitinib.Clin Pharmacokinet. 2005;44(11):1165-77. doi: 10.2165/00003088-200544110-00004. Clin Pharmacokinet. 2005. PMID: 16231967
-
Association of pharmacokinetics and pharmacogenomics with safety and efficacy of gefitinib in patients with EGFR mutation positive advanced non-small cell lung cancer.Lung Cancer. 2016 Mar;93:69-76. doi: 10.1016/j.lungcan.2016.01.005. Epub 2016 Jan 11. Lung Cancer. 2016. PMID: 26898617
-
Clinical pharmacokinetics of tyrosine kinase inhibitors: focus on 4-anilinoquinazolines.Clin Pharmacokinet. 2011 Jun;50(6):371-403. doi: 10.2165/11587020-000000000-00000. Clin Pharmacokinet. 2011. PMID: 21553932 Review.
-
Current knowledge and future directions of the selective epidermal growth factor receptor inhibitors erlotinib (Tarceva) and gefitinib (Iressa).Oncologist. 2005 Sep;10(8):579-89. doi: 10.1634/theoncologist.10-8-579. Oncologist. 2005. PMID: 16177282 Review.
Cited by
-
A Bottom-up Approach for Mutant and Wild Type Collies Using Physiologically Based Pharmacokinetic (PBPK) Modeling: A Case Study Using Loperamide.AAPS J. 2025 Jun 2;27(4):101. doi: 10.1208/s12248-025-01061-6. AAPS J. 2025. PMID: 40457124
-
An HNSCC syngeneic mouse model for tumor immunology research and preclinical evaluation.Int J Mol Med. 2020 Oct;46(4):1501-1513. doi: 10.3892/ijmm.2020.4680. Epub 2020 Jul 22. Int J Mol Med. 2020. PMID: 32700748 Free PMC article.
-
Drug Exposure to Establish Pharmacokinetic-Response Relationships in Oncology.Clin Pharmacokinet. 2020 Feb;59(2):123-135. doi: 10.1007/s40262-019-00828-3. Clin Pharmacokinet. 2020. PMID: 31654368 Review.
-
Physiologically based metformin pharmacokinetics model of mice and scale-up to humans for the estimation of concentrations in various tissues.PLoS One. 2021 Apr 7;16(4):e0249594. doi: 10.1371/journal.pone.0249594. eCollection 2021. PLoS One. 2021. PMID: 33826656 Free PMC article. Clinical Trial.
-
Use of physiological based pharmacokinetic modeling for cross-species prediction of pharmacokinetic and tissue distribution profiles of a novel niclosamide prodrug.Front Pharmacol. 2023 Apr 11;14:1099425. doi: 10.3389/fphar.2023.1099425. eCollection 2023. Front Pharmacol. 2023. PMID: 37113753 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

