Epstein-Barr Virus gp350 Can Functionally Replace the Rhesus Lymphocryptovirus Major Membrane Glycoprotein and Does Not Restrict Infection of Rhesus Macaques
- PMID: 26559839
- PMCID: PMC4719629
- DOI: 10.1128/JVI.02531-15
Epstein-Barr Virus gp350 Can Functionally Replace the Rhesus Lymphocryptovirus Major Membrane Glycoprotein and Does Not Restrict Infection of Rhesus Macaques
Abstract
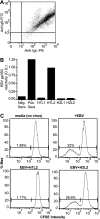
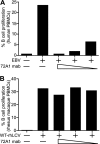
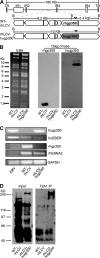
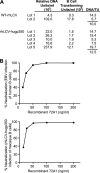
Primary Epstein-Barr virus (EBV) infection is the most common cause of infectious mononucleosis, and persistent infection is associated with multiple cancers. EBV vaccine development has focused on the major membrane glycoprotein, gp350, since it is the major target for antibodies that neutralize infection of B cells. However, EBV has tropism for both B cells and epithelial cells, and it is unknown whether serum neutralizing antibodies against B cell infection will provide sufficient protection against virus infection initiated at the oral mucosa. This could be stringently tested by passive antibody transfer and oral virus challenge in the rhesus macaque model for EBV infection. However, only neutralizing monoclonal antibodies (MAbs) against EBV are available, and EBV is unable to infect rhesus macaques because of a host range restriction with an unknown mechanism. We cloned the prototypic EBV-neutralizing antibody, 72A1, and found that recombinant 72A1 did not neutralize rhesus lymphocryptovirus (rhLCV) infection of macaque B cells. Therefore, we constructed a chimeric rhLCV in which the native major membrane glycoprotein was replaced with EBV gp350. This chimeric rhLCV became sensitive to neutralization by the 72A1 MAb, efficiently immortalized macaque B cells in vitro, and successfully established acute and persistent infection after oral inoculation of rhesus macaques. Thus, EBV gp350 can functionally replace rhLCV gp350 and does not restrict rhLCV infection in vitro or in vivo. The chimeric rhLCV enables direct use of an EBV-specific MAb to investigate the effects of serum neutralizing antibodies against B cell infection on oral viral challenge in rhesus macaques.
Importance: This study asked whether the EBV major membrane glycoprotein could functionally replace the rhLCV major membrane glycoprotein. We found that an rhLCV humanized with EBV gp350 is capable of efficiently immortalizing monkey B cells in vitro and reproduces acute and persistent infection after oral inoculation of macaques. These results advance our understanding of why EBV cannot infect rhesus macaques by proving that viral attachment through gp350 is not the mechanism for EBV host range restriction. Humanization of rhLCV with EBV gp350 also confers susceptibility to a potent EBV-neutralizing MAb and provides a novel and significant enhancement to the rhesus macaque animal model where both the clinical utility and biological role of neutralizing MAbs against B cell or epithelial cell infection can now be directly tested in the most accurate animal model for EBV infection.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures
Similar articles
-
Multivalent MVA-vectored vaccine elicits EBV neutralizing antibodies in rhesus macaques that reduce EBV infection in humanized mice.Front Immunol. 2024 Sep 13;15:1445209. doi: 10.3389/fimmu.2024.1445209. eCollection 2024. Front Immunol. 2024. PMID: 39346922 Free PMC article.
-
Soluble rhesus lymphocryptovirus gp350 protects against infection and reduces viral loads in animals that become infected with virus after challenge.PLoS Pathog. 2011 Oct;7(10):e1002308. doi: 10.1371/journal.ppat.1002308. Epub 2011 Oct 20. PLoS Pathog. 2011. PMID: 22028652 Free PMC article.
-
Vaccination with nanoparticles displaying gH/gL from Epstein-Barr virus elicits limited cross-protection against rhesus lymphocryptovirus.Cell Rep Med. 2024 Jun 18;5(6):101587. doi: 10.1016/j.xcrm.2024.101587. Epub 2024 May 22. Cell Rep Med. 2024. PMID: 38781964 Free PMC article.
-
Non-human Primate Lymphocryptoviruses: Past, Present, and Future.Curr Top Microbiol Immunol. 2015;391:385-405. doi: 10.1007/978-3-319-22834-1_13. Curr Top Microbiol Immunol. 2015. PMID: 26428382 Review.
-
A new animal model for Epstein-Barr virus pathogenesis.Curr Top Microbiol Immunol. 2001;258:201-19. doi: 10.1007/978-3-642-56515-1_13. Curr Top Microbiol Immunol. 2001. PMID: 11443863 Review. No abstract available.
Cited by
-
Mountain gorilla lymphocryptovirus has Epstein-Barr virus-like epidemiology and pathology in infants.Sci Rep. 2017 Jul 13;7(1):5352. doi: 10.1038/s41598-017-04877-1. Sci Rep. 2017. PMID: 28706209 Free PMC article.
-
Nonhuman primate models of human viral infections.Nat Rev Immunol. 2018 Jun;18(6):390-404. doi: 10.1038/s41577-018-0005-7. Nat Rev Immunol. 2018. PMID: 29556017 Free PMC article. Review.
-
Neutralizing Antibodies Protect against Oral Transmission of Lymphocryptovirus.Cell Rep Med. 2020 Jun 23;1(3):100033. doi: 10.1016/j.xcrm.2020.100033. Cell Rep Med. 2020. PMID: 32724901 Free PMC article.
-
Epstein-Barr virus BALF0/1 subverts the Caveolin and ERAD pathways to target B cell receptor complexes for degradation.Proc Natl Acad Sci U S A. 2025 Jan 28;122(4):e2400167122. doi: 10.1073/pnas.2400167122. Epub 2025 Jan 23. Proc Natl Acad Sci U S A. 2025. PMID: 39847318 Free PMC article.
-
Antibody Generation and Immunogenicity Analysis of EBV gp42 N-Terminal Region.Viruses. 2021 Nov 28;13(12):2380. doi: 10.3390/v13122380. Viruses. 2021. PMID: 34960650 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

