Eye Movement Deficits Are Consistent with a Staging Model of pTDP-43 Pathology in Amyotrophic Lateral Sclerosis
- PMID: 26559944
- PMCID: PMC4641606
- DOI: 10.1371/journal.pone.0142546
Eye Movement Deficits Are Consistent with a Staging Model of pTDP-43 Pathology in Amyotrophic Lateral Sclerosis
Abstract
Background: The neuropathological process underlying amyotrophic lateral sclerosis (ALS) can be traced as a four-stage progression scheme of sequential corticofugal axonal spread. The examination of eye movement control gains deep insights into brain network pathology and provides the opportunity to detect both disturbance of the brainstem oculomotor circuitry as well as executive deficits of oculomotor function associated with higher brain networks.
Objective: To study systematically oculomotor characteristics in ALS and its underlying network pathology in order to determine whether eye movement deterioration can be categorized within a staging system of oculomotor decline that corresponds to the neuropathological model.
Methods: Sixty-eight ALS patients and 31 controls underwent video-oculographic, clinical and neuropsychological assessments.
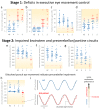
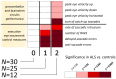
Results: Oculomotor examinations revealed increased anti- and delayed saccades' errors, gaze-palsy and a cerebellary type of smooth pursuit disturbance. The oculomotor disturbances occurred in a sequential manner: Stage 1, only executive control of eye movements was affected. Stage 2 indicates disturbed executive control plus 'genuine' oculomotor dysfunctions such as gaze-paly. We found high correlations (p<0.001) between the oculomotor stages and both, the clinical presentation as assessed by the ALS Functional Rating Scale (ALSFRS) score, and cognitive scores from the Edinburgh Cognitive and Behavioral ALS Screen (ECAS).
Conclusions: Dysfunction of eye movement control in ALS can be characterized by a two-staged sequential pattern comprising executive deficits in Stage 1 and additional impaired infratentorial oculomotor control pathways in Stage 2. This pattern parallels the neuropathological staging of ALS and may serve as a technical marker of the neuropathological spreading.
Conflict of interest statement
Figures
Similar articles
-
The multisystem degeneration amyotrophic lateral sclerosis - neuropathological staging and clinical translation.Arch Ital Biol. 2017 Dec 1;155(4):118-130. doi: 10.12871/00039829201746. Arch Ital Biol. 2017. PMID: 29405032 Review.
-
Spread of pathology in amyotrophic lateral sclerosis: assessment of phosphorylated TDP-43 along axonal pathways.Acta Neuropathol Commun. 2015 Jul 28;3:47. doi: 10.1186/s40478-015-0226-y. Acta Neuropathol Commun. 2015. PMID: 26216351 Free PMC article.
-
Multimodal in vivo staging in amyotrophic lateral sclerosis using artificial intelligence.Ann Clin Transl Neurol. 2022 Jul;9(7):1069-1079. doi: 10.1002/acn3.51601. Epub 2022 Jun 9. Ann Clin Transl Neurol. 2022. PMID: 35684940 Free PMC article.
-
Stages of pTDP-43 pathology in amyotrophic lateral sclerosis.Ann Neurol. 2013 Jul;74(1):20-38. doi: 10.1002/ana.23937. Epub 2013 Jun 19. Ann Neurol. 2013. PMID: 23686809 Free PMC article.
-
[Oculomotor disorders in patients with amyotrophic lateral sclerosis].Zh Nevrol Psikhiatr Im S S Korsakova. 2025;125(4):7-12. doi: 10.17116/jnevro20251250417. Zh Nevrol Psikhiatr Im S S Korsakova. 2025. PMID: 40350723 Review. Russian.
Cited by
-
Eye movement alterations in presymptomatic C9orf72 expansion gene carriers.J Neurol. 2021 Sep;268(9):3390-3399. doi: 10.1007/s00415-021-10510-z. Epub 2021 Mar 11. J Neurol. 2021. PMID: 33709219 Free PMC article.
-
Diagnostic value of video-oculography in progressive supranuclear palsy: a controlled study in 100 patients.J Neurol. 2021 Sep;268(9):3467-3475. doi: 10.1007/s00415-021-10522-9. Epub 2021 Mar 21. J Neurol. 2021. PMID: 33744980 Free PMC article.
-
Motor-Independent Cognitive Testing in Motor Degenerative Diseases.J Clin Med. 2022 Feb 3;11(3):814. doi: 10.3390/jcm11030814. J Clin Med. 2022. PMID: 35160265 Free PMC article.
-
Disease-modifying and symptomatic treatment of amyotrophic lateral sclerosis.Ther Adv Neurol Disord. 2017 Oct 9;11:1756285617734734. doi: 10.1177/1756285617734734. eCollection 2018. Ther Adv Neurol Disord. 2017. PMID: 29399045 Free PMC article. Review.
-
Functional connectivity changes resemble patterns of pTDP-43 pathology in amyotrophic lateral sclerosis.Sci Rep. 2016 Dec 8;6:38391. doi: 10.1038/srep38391. Sci Rep. 2016. PMID: 27929102 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

