Ca(2+) leakage out of the sarcoplasmic reticulum is increased in type I skeletal muscle fibres in aged humans
- PMID: 26574292
- PMCID: PMC4713735
- DOI: 10.1113/JP271382
Ca(2+) leakage out of the sarcoplasmic reticulum is increased in type I skeletal muscle fibres in aged humans
Abstract
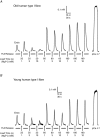
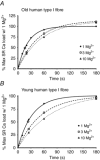
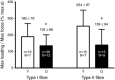
Key points: The amount of Ca(2+) stored in the sarcoplasmic reticulum (SR) of muscle fibres is decreased in aged individuals, and an important question is whether this results from increased Ca(2+) leakage out through the Ca(2+) release channels (ryanodine receptors; RyRs). The present study examined the effects of blocking the RyRs with Mg(2+), or applying a strong reducing treatment, on net Ca(2+) accumulation by the SR in skinned muscle fibres from Old (∼70 years) and Young (∼24 years) adults. Raising cytoplasmic [Mg(2+)] and reducing treatment increased net SR Ca(2+) accumulation in type I fibres of Old subjects relative to that in Young. The densities of RyRs and dihydropyridine receptors were not significantly changed in the muscle of Old subjects. These findings indicate that oxidative modification of the RyRs causes increased Ca(2+) leakage from the SR in muscle fibres in Old subjects, which probably deleteriously affects normal muscle function both directly and indirectly.
Abstract: The present study examined whether the lower Ca(2+) storage levels in the sarcoplasmic reticulum (SR) in vastus lateralis muscle fibres in Old (70 ± 4 years) relative to Young (24 ± 4 years) human subjects is the result of increased leakage of Ca(2+) out of the SR through the Ca(2+) release channels/ryanodine receptors (RyRs) and due to oxidative modification of the RyRs. SR Ca(2+) accumulation in mechanically skinned muscle fibres was examined in the presence of 1, 3 or 10 mm cytoplasmic Mg(2+) because raising [Mg(2+)] strongly inhibits Ca(2+) efflux through the RyRs. In type I fibres of Old subjects, SR Ca(2+) accumulation in the presence of 1 mm Mg(2+) approached saturation at shorter loading times than in Young subjects, consistent with Ca(2+) leakage limiting net uptake, and raising [Mg(2+)] to 10 mm in such fibres increased maximal SR Ca(2+) accumulation. No significant differences were seen in type II fibres. Treatment with dithiothreitol (10 mm for 5 min), a strong reducing agent, also increased maximal SR Ca(2+) accumulation at 1 mm Mg(2+) in type I fibres of Old subjects but not in other fibres. The densities of dihydropyridine receptors and RyRs were not significantly different in muscles of Old relative to Young subjects. These findings indicate that Ca(2+) leakage from the SR is increased in type I fibres in Old subjects by reversible oxidative modification of the RyRs; this increased SR Ca(2+) leak is expected to have both direct and indirect deleterious effects on Ca(2+) movements and muscle function.
© 2015 The Authors. The Journal of Physiology © 2015 The Physiological Society.
Figures





Similar articles
-
Contractile properties and sarcoplasmic reticulum calcium content in type I and type II skeletal muscle fibres in active aged humans.J Physiol. 2015 Jun 1;593(11):2499-514. doi: 10.1113/JP270179. Epub 2015 Apr 17. J Physiol. 2015. PMID: 25809942 Free PMC article.
-
Sarcoplasmic reticulum Ca2+ uptake and leak properties, and SERCA isoform expression, in type I and type II fibres of human skeletal muscle.J Physiol. 2014 Mar 15;592(6):1381-95. doi: 10.1113/jphysiol.2013.269373. Epub 2014 Jan 27. J Physiol. 2014. PMID: 24469076 Free PMC article.
-
Orthograde signal of dihydropyridine receptor increases Ca2+ leakage after repeated contractions in rat fast-twitch muscles in vivo.Am J Physiol Cell Physiol. 2021 May 1;320(5):C806-C821. doi: 10.1152/ajpcell.00364.2020. Epub 2021 Feb 17. Am J Physiol Cell Physiol. 2021. PMID: 33596151
-
The Ca 2+ leak paradox and rogue ryanodine receptors: SR Ca 2+ efflux theory and practice.Prog Biophys Mol Biol. 2006 Jan-Apr;90(1-3):172-85. doi: 10.1016/j.pbiomolbio.2005.06.010. Epub 2005 Jul 18. Prog Biophys Mol Biol. 2006. PMID: 16326215 Free PMC article. Review.
-
Ca2+ stores regulate ryanodine receptor Ca2+ release channels via luminal and cytosolic Ca2+ sites.Clin Exp Pharmacol Physiol. 2007 Sep;34(9):889-96. doi: 10.1111/j.1440-1681.2007.04708.x. Clin Exp Pharmacol Physiol. 2007. PMID: 17645636 Review.
Cited by
-
Fast skeletal muscle troponin activator CK-2066260 mitigates skeletal muscle weakness independently of the underlying cause.J Cachexia Sarcopenia Muscle. 2020 Dec;11(6):1747-1757. doi: 10.1002/jcsm.12624. Epub 2020 Sep 21. J Cachexia Sarcopenia Muscle. 2020. PMID: 32954682 Free PMC article.
-
Store-operated Ca2+ entry is activated by every action potential in skeletal muscle.Commun Biol. 2018 Apr 19;1:31. doi: 10.1038/s42003-018-0033-7. eCollection 2018. Commun Biol. 2018. PMID: 30271917 Free PMC article.
-
Numb is required for optimal contraction of skeletal muscle.J Cachexia Sarcopenia Muscle. 2022 Feb;13(1):454-466. doi: 10.1002/jcsm.12907. Epub 2022 Jan 9. J Cachexia Sarcopenia Muscle. 2022. PMID: 35001540 Free PMC article.
-
Caffeine as a tool to investigate sarcoplasmic reticulum and intracellular calcium dynamics in human skeletal muscles.J Muscle Res Cell Motil. 2021 Jun;42(2):281-289. doi: 10.1007/s10974-020-09574-7. Epub 2020 Feb 7. J Muscle Res Cell Motil. 2021. PMID: 32034582 Review.
-
Senescence Is Associated With Elevated Intracellular Resting [Ca2 +] in Mice Skeletal Muscle Fibers. An in vivo Study.Front Physiol. 2021 Jan 12;11:601189. doi: 10.3389/fphys.2020.601189. eCollection 2020. Front Physiol. 2021. PMID: 33510646 Free PMC article.
References
-
- Aracena‐Parks P, Goonasekera SA, Gilman CP, Dirksen RT, Hidalgo C & Hamilton SL (2006). Identification of cysteines involved in S‐nitrosylation, S‐glutathionylation, and oxidation to disulfides in ryanodine receptor type 1. J Biol Chem 281, 40354–40368. - PubMed
-
- Aracena P, Sanchez G, Donoso P, Hamilton SL & Hidalgo C (2003). S‐glutathionylation decreases Mg2+ inhibition and S‐nitrosylation enhances Ca2+ activation of RyR1 channels. J Biol Chem 278, 42927–42935. - PubMed
-
- Ballak SB, Degens H, de Haan A & Jaspers RT (2014). Aging related changes in determinants of muscle force generating capacity: a comparison of muscle aging in men and male rodents. Ageing Res Rev 14, 43–55. - PubMed
-
- Bottinelli R & Reggiani C (2000). Human skeletal muscle fibres: molecular and functional diversity. Prog Biophys Mol Biol 73, 195–262. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

