MALDI Imaging Mass Spectrometry Spatially Maps Age-Related Deamidation and Truncation of Human Lens Aquaporin-0
- PMID: 26574799
- PMCID: PMC4675204
- DOI: 10.1167/iovs.15-18117
MALDI Imaging Mass Spectrometry Spatially Maps Age-Related Deamidation and Truncation of Human Lens Aquaporin-0
Abstract
Purpose: To spatially map human lens Aquaporin-0 (AQP0) protein modifications, including lipidation, truncation, and deamidation, from birth through middle age using matrix-assisted laser desorption ionization (MALDI) imaging mass spectrometry (IMS).
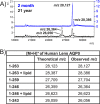
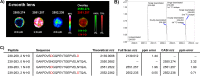
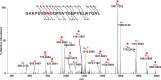
Methods: Human lens sections were water-washed to facilitate detection of membrane protein AQP0. We acquired MALDI images from eight human lenses ranging in age from 2 months to 63 years. In situ tryptic digestion was used to generate peptides of AQP0 and peptide images were acquired on a 15T Fourier transform ion cyclotron resonance (FTICR) mass spectrometer. Peptide extracts were analyzed by liquid chromatography-tandem mass spectrometry (LC-MS/MS) and database searched to identify peptides observed in MALDI imaging experiments.
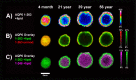
Results: Unmodified, truncated, and fatty acid-acylated forms of AQP0 were detected in protein imaging experiments. Full-length AQP0 was fatty acid acylated in the core and cortex of young (2- and 4-month) lenses. Acylated and unmodified AQP0 were C-terminally truncated in older lens cores. Deamidated tryptic peptides (+0.9847 Da) were mass resolved from unmodified peptides by FTICR MS. Peptide images revealed differential localization of un-, singly-, and doubly-deamidated AQP0 C-terminal peptide (239-263). Deamidation was present at 4 months and increases with age. Liquid chromatography-MS/MS results indicated N246 undergoes deamidation more rapidly than N259.
Conclusions: Results indicated AQP0 fatty acid acylation and deamidation occur during early development. Progressive age-related AQP0 processing, including deamidation and truncation, was mapped in human lenses as a function of age. The localization of these modified AQP0 forms suggests where AQP0 functions may change throughout lens development and aging.
Figures


Similar articles
-
Spatial analysis of human lens aquaporin-0 post-translational modifications by MALDI mass spectrometry tissue profiling.Exp Eye Res. 2011 Dec;93(6):912-20. doi: 10.1016/j.exer.2011.10.007. Epub 2011 Oct 25. Exp Eye Res. 2011. PMID: 22036630 Free PMC article.
-
Post-translational modifications of aquaporin 0 (AQP0) in the normal human lens: spatial and temporal occurrence.Biochemistry. 2004 Aug 3;43(30):9856-65. doi: 10.1021/bi0496034. Biochemistry. 2004. PMID: 15274640
-
Water permeability of C-terminally truncated aquaporin 0 (AQP0 1-243) observed in the aging human lens.Invest Ophthalmol Vis Sci. 2003 Nov;44(11):4820-8. doi: 10.1167/iovs.02-1317. Invest Ophthalmol Vis Sci. 2003. PMID: 14578404
-
Structural function of MIP/aquaporin 0 in the eye lens; genetic defects lead to congenital inherited cataracts.Handb Exp Pharmacol. 2009;(190):265-97. doi: 10.1007/978-3-540-79885-9_14. Handb Exp Pharmacol. 2009. PMID: 19096783 Review.
-
Spatiotemporal changes in the human lens proteome: Critical insights into long-lived proteins.Prog Retin Eye Res. 2020 May;76:100802. doi: 10.1016/j.preteyeres.2019.100802. Epub 2019 Nov 6. Prog Retin Eye Res. 2020. PMID: 31704338 Free PMC article. Review.
Cited by
-
Dynamic Range Expansion by Gas-Phase Ion Fractionation and Enrichment for Imaging Mass Spectrometry.Anal Chem. 2020 Oct 6;92(19):13092-13100. doi: 10.1021/acs.analchem.0c02121. Epub 2020 Sep 9. Anal Chem. 2020. PMID: 32845133 Free PMC article.
-
Acceleration of age-induced proteolysis in the guinea pig lens nucleus by in vivo exposure to hyperbaric oxygen: A mass spectrometry analysis.Exp Eye Res. 2021 Sep;210:108697. doi: 10.1016/j.exer.2021.108697. Epub 2021 Jul 4. Exp Eye Res. 2021. PMID: 34233175 Free PMC article.
-
Data-Independent Acquisition Mass Spectrometry of the Human Lens Enhances Spatiotemporal Measurement of Fiber Cell Aging.J Am Soc Mass Spectrom. 2021 Dec 1;32(12):2755-2765. doi: 10.1021/jasms.1c00193. Epub 2021 Oct 27. J Am Soc Mass Spectrom. 2021. PMID: 34705440 Free PMC article.
-
Unsupervised Structural Classification of Dissolved Organic Matter Based on Fragmentation Pathways.Environ Sci Technol. 2022 Jan 18;56(2):1458-1468. doi: 10.1021/acs.est.1c04726. Epub 2022 Jan 4. Environ Sci Technol. 2022. PMID: 34981937 Free PMC article.
-
Lens Aquaporins in Health and Disease: Location is Everything!Front Physiol. 2022 Apr 19;13:882550. doi: 10.3389/fphys.2022.882550. eCollection 2022. Front Physiol. 2022. PMID: 35514349 Free PMC article. Review.
References
-
- Mathias RT,, Rae JL,, Baldo GJ. Physiological properties of the normal lens. Physiol Rev. 1997; 77: 21–50. - PubMed
-
- Jacob TJ. The relationship between cataract cell swelling and volume regulation. Prog Retin Eye Res. 1999; 18: 223–233. - PubMed
-
- Taylor VL,, al-Ghoul KJ,, Lane CW,, Davis VA,, Kuszak JR,, Costello MJ. Morphology of the normal human lens. Invest Ophthalmol Vis Sci. 1996; 37: 1396–1410. - PubMed
-
- Bassnett S. Lens organelle degradation. Exp Eye Res. 2002; 74: 1–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 5P41-GM103391-05/GM/NIGMS NIH HHS/United States
- S10RR027714/RR/NCRR NIH HHS/United States
- P30-EY08126/EY/NEI NIH HHS/United States
- P41 GM103391/GM/NIGMS NIH HHS/United States
- 3U42OD011158/OD/NIH HHS/United States
- 1S10OD012359-01/OD/NIH HHS/United States
- R01 EY013462/EY/NEI NIH HHS/United States
- EY-13462/EY/NEI NIH HHS/United States
- P30 EY008126/EY/NEI NIH HHS/United States
- S10 OD012359/OD/NIH HHS/United States
- T32-EY07135/EY/NEI NIH HHS/United States
- S10 RR027714/RR/NCRR NIH HHS/United States
- U42 OD011158/OD/NIH HHS/United States
- T32 EY007135/EY/NEI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

