Cancer metastases: challenges and opportunities
- PMID: 26579471
- PMCID: PMC4629446
- DOI: 10.1016/j.apsb.2015.07.005
Cancer metastases: challenges and opportunities
Abstract
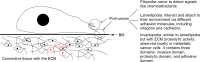
Cancer metastasis is the major cause of cancer morbidity and mortality, and accounts for about 90% of cancer deaths. Although cancer survival rate has been significantly improved over the years, the improvement is primarily due to early diagnosis and cancer growth inhibition. Limited progress has been made in the treatment of cancer metastasis due to various factors. Current treatments for cancer metastasis are mainly chemotherapy and radiotherapy, though the new generation anti-cancer drugs (predominantly neutralizing antibodies for growth factors and small molecule kinase inhibitors) do have the effects on cancer metastasis in addition to their effects on cancer growth. Cancer metastasis begins with detachment of metastatic cells from the primary tumor, travel of the cells to different sites through blood/lymphatic vessels, settlement and growth of the cells at a distal site. During the process, metastatic cells go through detachment, migration, invasion and adhesion. These four essential, metastatic steps are inter-related and affected by multi-biochemical events and parameters. Additionally, it is known that tumor microenvironment (such as extracellular matrix structure, growth factors, chemokines, matrix metalloproteinases) plays a significant role in cancer metastasis. The biochemical events and parameters involved in the metastatic process and tumor microenvironment have been targeted or can be potential targets for metastasis prevention and inhibition. This review provides an overview of these metastasis essential steps, related biochemical factors, and targets for intervention.
Keywords: Adhesion; BM, basement membrane; CAFs, cancer-associated fibroblasts; CAMs, cell adhesion molecules; CAT, collective amoeboid transition; CCL2, chemokine (C–C motif) ligand 2; CCR3, chemokine receptor 3; COX2, cyclooxygenase 2; CSF-1, chemokine colonystimulating factor–1; CTGF, connective tissue growth factor; CXCR2, chemokine receptor type 2; Cancer; Col, collagen; DISC, death-inducing signaling complex; Detachment; ECM, extracellular matrix; EGF, epidermal growth factor; EGFR, EGF receptor; EMT, epithelial–mesenchymal transition; FAK, focal adhesion kinase; FAs, focal adhesions; FGF, fibroblast growth factor; FN, fibronectin; HA, hyaluronan; HGF, hepatocyte growth factor; HIFs, hypoxia-inducible factors; IKK, IκB kinase; Invasion; JAK, the Janus kinases; LN, laminin; MAPK, mitogen-activated protein kinase; MAT, mesenchymal to amoeboid transition; MET, mesenchymal–epithelial transition; MMPs, matrix metalloproteinases; Metastasis; Migration; PDGF, platelet-derived growth factor; PI3K, phosphatidylinositol 3-kinase; STATs, signal transducers and activators of transcription; TAMs, tumor-associated macrophages; TGF-β, transforming growth factor β; TME, tumor microenvironment; VCAMs, vascular cell adhesion molecules; VEGF, vascular endothelial growth factor; VN, vitronectin.
Figures










References
-
- Damsky WE, Theodosakis N, Bosenberg M. Melanoma metastasis: new concepts and evolving paradigms. Oncogene. 2014;33:2413–2422. - PubMed
-
- Weber GF. Why does cancer therapy lack effective anti-metastasis drugs? Cancer Lett. 2013;328:207–211. - PubMed
-
- Hu YY, Zheng MH, Zhang R, Liang YM, Han H. Notch signaling pathway and cancer metastasis. Adv Exp Med Biol. 2012;727:186–198. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

