Role of necroptosis in the pathogenesis of solid organ injury
- PMID: 26583318
- PMCID: PMC4670925
- DOI: 10.1038/cddis.2015.316
Role of necroptosis in the pathogenesis of solid organ injury
Abstract
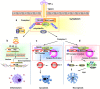
Necroptosis is a type of regulated cell death dependent on the activity of receptor-interacting serine/threonine-protein (RIP) kinases. However, unlike apoptosis, it is caspase independent. Increasing evidence has implicated necroptosis in the pathogenesis of disease, including ischemic injury, neurodegeneration, viral infection and many others. Key players of the necroptosis signalling pathway are now widely recognized as therapeutic targets. Necrostatins may be developed as potent inhibitors of necroptosis, targeting the activity of RIPK1. Necrostatin-1, the first generation of necrostatins, has been shown to confer potent protective effects in different animal models. This review will summarize novel insights into the involvement of necroptosis in specific injury of different organs, and the therapeutic platform that it provides for treatment.
Figures


References
-
- 1Vandenabeele P, Galluzzi L, Vanden Berghe T, Kroemer G. Molecular mechanisms of necroptosis: an ordered cellular explosion. Nat Rev Mol Cell Biol 2010; 11: 700–714. - PubMed
-
- 2Kanduc D, Mittelman A, Serpico R, Sinigaglia E, Sinha AA, Natale C et al. Cell death: apoptosis versus necrosis (review). Int J Oncol 2002; 21: 165–170. - PubMed
-
- 3Newton K, Dugger DL, Wickliffe KE, Kapoor N, de Almagro MC, Vucic D et al. Activity of protein kinase RIPK3 determines whether cells die by necroptosis or apoptosis. Science 2014; 343: 1357–1360. - PubMed
-
- 4Sun L, Wang H, Wang Z, He S, Chen S, Liao D et al. Mixed lineage kinase domain-like protein mediates necrosis signaling downstream of RIP3 kinase. Cell 2012; 148: 213–227. - PubMed
-
- 5Luedde M, Lutz M, Carter N, Sosna J, Jacoby C, Vucur M et al. RIP3, a kinase promoting necroptotic cell death, mediates adverse remodelling after myocardial infarction. Cardiovasc Res 2014; 103: 206–216. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

