Fulminant demyelinating encephalomyelitis: Insights from antibody studies and neuropathology
- PMID: 26587556
- PMCID: PMC4635550
- DOI: 10.1212/NXI.0000000000000175
Fulminant demyelinating encephalomyelitis: Insights from antibody studies and neuropathology
Abstract
Objectives: Antibodies to myelin oligodendrocyte glycoprotein (MOG) are detectable in inflammatory demyelinating CNS diseases, and MOG antibody-associated diseases seem to have a better prognosis despite occasionally severe presentations.
Methods: We report the case of a 71-year-old patient with acute visual and gait disturbance that dramatically worsened to bilateral amaurosis, tetraplegia, and respiratory insufficiency within a few days.
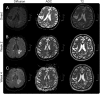
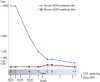
Results: MRI showed multiple progressive cerebral and spinal lesions with diffusion restriction (including both optic nerves) and marginal contrast enhancement. Routine blood and CSF measures including oligoclonal bands were normal. At disease onset, MOG immunoglobulin G was detected (serum titer 1:1,280, corresponding CSF titer was 1:20) and remained positive in patient serum. Aquaporin-4 antibodies were absent at disease onset but seroconverted to positive at week 9. In addition, CSF glial fibrillary acid protein and myelin basic protein levels were very high at onset but decreased during disease course. After 4 months, the patient died despite immunomodulatory treatment. Postmortem neuropathologic examination revealed an acute multiple sclerosis (MS) defined by multiple demyelinating lesions with a pronounced destructive component and loss of astrocytes. Lesion pattern of optic chiasm met MS pattern II characterized by antibody and complement-mediated demyelination.
Conclusion: The case with the clinical presentation of an acute demyelinating encephalomyelitis with predominant optic and spinal involvement, absent oligoclonal bands, a histopathology of acute MS pattern II and development of aquaporin-4 antibodies extends the spectrum of MOG antibody-associated encephalomyelitis. Although, MOG antibodies are suspected to indicate a favorable prognosis, fulminant disease courses are possible and warrant an aggressive immunotherapy.
Figures


References
-
- Karussis D. The diagnosis of multiple sclerosis and the various related demyelinating syndromes: a critical review. J Autoimmun 2014;48–49:134–142. - PubMed
-
- Lennon VA, Wingerchuk DM, Kryzer TJ, et al. A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet 2004;364:2106–2112. - PubMed
-
- Popescu BF, Lucchinetti CF. Pathology of demyelinating diseases. Annu Rev Pathol 2012;7:185–217. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
