The supramammillary nucleus and the claustrum activate the cortex during REM sleep
- PMID: 26601158
- PMCID: PMC4640625
- DOI: 10.1126/sciadv.1400177
The supramammillary nucleus and the claustrum activate the cortex during REM sleep
Abstract
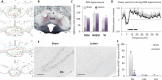
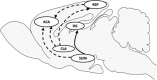
Evidence in humans suggests that limbic cortices are more active during rapid eye movement (REM or paradoxical) sleep than during waking, a phenomenon fitting with the presence of vivid dreaming during this state. In that context, it seemed essential to determine which populations of cortical neurons are activated during REM sleep. Our aim in the present study is to fill this gap by combining gene expression analysis, functional neuroanatomy, and neurochemical lesions in rats. We find in rats that, during REM sleep hypersomnia compared to control and REM sleep deprivation, the dentate gyrus, claustrum, cortical amygdaloid nucleus, and medial entorhinal and retrosplenial cortices are the only cortical structures containing neurons with an increased expression of Bdnf, FOS, and ARC, known markers of activation and/or synaptic plasticity. Further, the dentate gyrus is the only cortical structure containing more FOS-labeled neurons during REM sleep hypersomnia than during waking. Combining FOS staining, retrograde labeling, and neurochemical lesion, we then provide evidence that FOS overexpression occurring in the cortex during REM sleep hypersomnia is due to projections from the supramammillary nucleus and the claustrum. Our results strongly suggest that only a subset of cortical and hippocampal neurons are activated and display plasticity during REM sleep by means of ascending projections from the claustrum and the supramammillary nucleus. Our results pave the way for future studies to identify the function of REM sleep with regard to dreaming and emotional memory processing.
Keywords: REM sleep; dentate gyrus; hippocampus; immediate early genes; supramammillary nucleus.
Figures
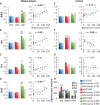
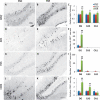

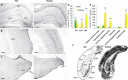
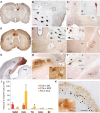
References
-
- Dement W. C., Kleitman N., Cyclic variations of EEG during sleep and their relation to eye movement, body motility, and dreaming. Electroencephalogr. Clin. Neurophysiol. 9, 673–690 (1957). - PubMed
-
- Maloney K. J., Cape E. G., Gotman J., Jones B. E., High-frequency γ electroencephalogram activity in association with sleep-wake states and spontaneous behaviors in the rat. Neuroscience 76, 541–555 (1997). - PubMed
-
- Maquet P., Péters J., Aerts J., Delfiore G., Degueldre C., Luxen A., Franck G., Functional neuroanatomy of human rapid-eye-movement sleep and dreaming. Nature 383, 163–166 (1996). - PubMed
-
- Steriade M., Timofeev I., Grenier F., Natural waking and sleep states: A view from inside neocortical neurons. J. Neurophysiol. 85, 1969–1985 (2001). - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

