Uncoupling Caveolae From Intracellular Signaling In Vivo
- PMID: 26602865
- PMCID: PMC4740205
- DOI: 10.1161/CIRCRESAHA.115.307767
Uncoupling Caveolae From Intracellular Signaling In Vivo
Abstract
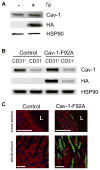
Rationale: Caveolin-1 (Cav-1) negatively regulates endothelial nitric oxide (NO) synthase-derived NO production, and this has been mapped to several residues on Cav-1, including F92. Herein, we reasoned that endothelial expression of an F92ACav-1 transgene would let us decipher the mechanisms and relationships between caveolae structure and intracellular signaling.
Objective: This study was designed to separate caveolae formation from its downstream signaling effects.
Methods and results: An endothelial-specific doxycycline-regulated mouse model for the expression of Cav-1-F92A was developed. Blood pressure by telemetry and nitric oxide bioavailability by electron paramagnetic resonance and phosphorylation of vasodilator-stimulated phosphoprotein were determined. Caveolae integrity in the presence of Cav-1-F92A was measured by stabilization of caveolin-2, sucrose gradient, and electron microscopy. Histological analysis of heart and lung, echocardiography, and signaling were performed.
Conclusions: This study shows that mutant Cav-1-F92A forms caveolae structures similar to WT but leads to increases in NO bioavailability in vivo, thereby demonstrating that caveolae formation and downstream signaling events occur through independent mechanisms.
Keywords: caveolin-1; cell endothelial cell; eNOS; mice; nitric oxide; vascular function.
© 2015 American Heart Association, Inc.
Conflict of interest statement
The authors declare that there are no conflicts of interest. The sponsors had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Figures



References
-
- Frank PG, Pavlides S, Lisanti MP. Caveolae and transcytosis in endothelial cells: Role in atherosclerosis. Cell and tissue research. 2009;335:41–47. - PubMed
-
- Gratton J-P, Bernatchez P, Sessa WC. Caveolae and caveolins in the cardiovascular system. Circulation Research. 2004;94:1408–1417. - PubMed
-
- Parton RG, Simons K. The multiple faces of caveolae. Nat Rev Mol Cell Biol. 2007;8:185–194. - PubMed
-
- Drab M, Verkade P, Elger M, Kasper M, Lohn M, Lauterbach B, Menne J, Lindschau C, Mende F, Luft FC, Schedl A, Haller H, Kurzchalia TV. Loss of caveolae, vascular dysfunction, and pulmonary defects in caveolin-1 gene-disrupted mice. Science. 2001;293:2449–2452. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 HL61371/HL/NHLBI NIH HHS/United States
- R01 HL64793/HL/NHLBI NIH HHS/United States
- P30 DK079310/DK/NIDDK NIH HHS/United States
- R01 HL064793/HL/NHLBI NIH HHS/United States
- P30 CA023108/CA/NCI NIH HHS/United States
- R37 HL061371/HL/NHLBI NIH HHS/United States
- R01 HL057665/HL/NHLBI NIH HHS/United States
- R01 HL133018/HL/NHLBI NIH HHS/United States
- P01 HL1070295/HL/NHLBI NIH HHS/United States
- F32 HL119147/HL/NHLBI NIH HHS/United States
- R01 HL081190/HL/NHLBI NIH HHS/United States
- R01 HL061371/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

