Strict experimental evidence that apo-chaperonin GroEL does not accelerate protein folding, although it does accelerate one of its steps
- PMID: 26604318
- PMCID: PMC4687534
- DOI: 10.1073/pnas.1517712112
Strict experimental evidence that apo-chaperonin GroEL does not accelerate protein folding, although it does accelerate one of its steps
Conflict of interest statement
The authors declare no conflict of interest.
Figures
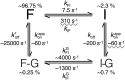
Comment in
-
Reply to Marchenko et al.: Flux analysis of GroEL-assisted protein folding/unfolding.Proc Natl Acad Sci U S A. 2015 Dec 15;112(50):E6833-4. doi: 10.1073/pnas.1520474112. Epub 2015 Nov 24. Proc Natl Acad Sci U S A. 2015. PMID: 26604310 Free PMC article. No abstract available.
Comment on
-
Intrinsic unfoldase/foldase activity of the chaperonin GroEL directly demonstrated using multinuclear relaxation-based NMR.Proc Natl Acad Sci U S A. 2015 Jul 21;112(29):8817-23. doi: 10.1073/pnas.1510083112. Epub 2015 Jun 29. Proc Natl Acad Sci U S A. 2015. PMID: 26124125 Free PMC article.
References
-
- Becker R, Döring W. Kinetische Behandlung der Keimbildung in übersättigten Dämpfen. Ann Phys. 1935;416(8):719–752.
-
- Ferrone F. Analysis of protein aggregation kinetics. Methods Enzymol. 1999;309:256–274. - PubMed
-
- Finkelstein AV. Time to overcome the high, long, and bumpy free energy barrier in a multi-stage process: The generalized steady-state approach. J Phys Chem B. 2015;119(1):158–163. - PubMed
-
- Marchenkov VV, et al. The interaction of the GroEL chaperone with early kinetic intermediates of renaturing proteins inhibits the formation of their native structure. Biophysics. 2004;49(6):888–894. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

