Does Gleason score at initial diagnosis predict efficacy of abiraterone acetate therapy in patients with metastatic castration-resistant prostate cancer? An analysis of abiraterone acetate phase III trials
- PMID: 26609008
- PMCID: PMC6279107
- DOI: 10.1093/annonc/mdv545
Does Gleason score at initial diagnosis predict efficacy of abiraterone acetate therapy in patients with metastatic castration-resistant prostate cancer? An analysis of abiraterone acetate phase III trials
Abstract
Background: The usefulness of Gleason score (<8 or ≥8) at initial diagnosis as a predictive marker of response to abiraterone acetate (AA) plus prednisone in patients with metastatic castration-resistant prostate cancer (mCRPC) was explored retrospectively.
Patients and methods: Initial diagnosis Gleason score was obtained in 1048 of 1195 (COU-AA-301, post-docetaxel) and 996 of 1088 (COU-AA-302, chemotherapy-naïve) patients treated with AA 1 g plus prednisone 5 mg twice daily by mouth or placebo plus prednisone. Efficacy end points included radiographic progression-free survival (rPFS) and overall survival (OS). Distributions and medians were estimated by Kaplan-Meier method and hazard ratio (HR) and 95% confidence interval (CI) by Cox model.
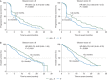
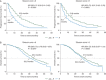
Results: Baseline characteristics were similar across studies and treatment groups. Regardless of Gleason score, AA treatment significantly improved rPFS in post-docetaxel [Gleason score <8: median, 6.4 versus 5.5 months (HR = 0.70; 95% CI 0.56-0.86), P = 0.0009 and Gleason score ≥8: median, 5.6 versus 2.9 months (HR = 0.58; 95% CI 0.48-0.72), P < 0.0001] and chemotherapy-naïve patients [Gleason score <8: median, 16.5 versus 8.2 months (HR = 0.50; 95% CI 0.40-0.62), P < 0.0001 and Gleason score ≥8: median, 13.8 versus 8.2 months (HR = 0.61; 95% CI 0.49-0.76), P < 0.0001]. Clinical benefit of AA treatment was also observed for OS, prostate-specific antigen (PSA) response, objective response and time to PSA progression across studies and Gleason score subgroups.
Conclusion: OS and rPFS trends demonstrate AA treatment benefit in patients with pre- or post-chemotherapy mCRPC regardless of Gleason score at initial diagnosis. The initial diagnostic Gleason score in patients with mCRPC should not be considered in the decision to treat with AA, as tumour metastases may no longer reflect the histology at the time of diagnosis.
Clinical trials number: COU-AA-301 (NCT00638690); COU-AA-302 (NCT00887198).
Keywords: Gleason score; abiraterone acetate; chemotherapy-naïve; post-chemotherapy; prostate cancer.
© The Author 2015. Published by Oxford University Press on behalf of the European Society for Medical Oncology. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Figures
Comment in
-
Diagnostic Gleason score and castration-resistant prostate cancer.Ann Oncol. 2016 Jun;27(6):962-964. doi: 10.1093/annonc/mdw179. Epub 2016 Apr 18. Ann Oncol. 2016. PMID: 27091809 No abstract available.
-
Is the Gleason score the driver for the treatment decision-making of patients with castration-resistant prostate cancer in the new era of the anti-androgenic therapies?Ann Oncol. 2016 Nov;27(11):2131-2133. doi: 10.1093/annonc/mdw283. Epub 2016 Aug 8. Ann Oncol. 2016. PMID: 27502707 No abstract available.
Similar articles
-
The Phase 3 COU-AA-302 Study of Abiraterone Acetate Plus Prednisone in Men with Chemotherapy-naïve Metastatic Castration-resistant Prostate Cancer: Stratified Analysis Based on Pain, Prostate-specific Antigen, and Gleason Score.Eur Urol. 2018 Jul;74(1):17-23. doi: 10.1016/j.eururo.2017.08.035. Epub 2017 Sep 20. Eur Urol. 2018. PMID: 28939004 Clinical Trial.
-
Updated interim efficacy analysis and long-term safety of abiraterone acetate in metastatic castration-resistant prostate cancer patients without prior chemotherapy (COU-AA-302).Eur Urol. 2014 Nov;66(5):815-25. doi: 10.1016/j.eururo.2014.02.056. Epub 2014 Mar 6. Eur Urol. 2014. PMID: 24647231 Free PMC article. Clinical Trial.
-
Prior Endocrine Therapy Impact on Abiraterone Acetate Clinical Efficacy in Metastatic Castration-resistant Prostate Cancer: Post-hoc Analysis of Randomised Phase 3 Studies.Eur Urol. 2016 May;69(5):924-32. doi: 10.1016/j.eururo.2015.10.021. Epub 2015 Oct 24. Eur Urol. 2016. PMID: 26508309 Free PMC article. Clinical Trial.
-
Matching-adjusted indirect comparison of talazoparib plus enzalutamide versus abiraterone acetate and docetaxel in mCRPC.Future Oncol. 2025 Apr;21(9):1075-1084. doi: 10.1080/14796694.2025.2471200. Epub 2025 Mar 5. Future Oncol. 2025. PMID: 40045559 Free PMC article.
-
Abiraterone Acetate for the Treatment of Chemotherapy-Naïve Metastatic Castration-Resistant Prostate Cancer: An Evidence Review Group Perspective of an NICE Single Technology Appraisal.Pharmacoeconomics. 2017 Feb;35(2):191-202. doi: 10.1007/s40273-016-0445-5. Pharmacoeconomics. 2017. PMID: 27566699 Free PMC article. Review.
Cited by
-
Predictors of long-term response to abiraterone in patients with metastastic castration-resistant prostate cancer: a retrospective cohort study.Oncotarget. 2016 Jun 28;7(26):40085-40094. doi: 10.18632/oncotarget.9485. Oncotarget. 2016. PMID: 27223078 Free PMC article.
-
Common Genetic Variation in CYP17A1 and Response to Abiraterone Acetate in Patients with Metastatic Castration-Resistant Prostate Cancer.Int J Mol Sci. 2016 Jul 9;17(7):1097. doi: 10.3390/ijms17071097. Int J Mol Sci. 2016. PMID: 27409606 Free PMC article.
-
Predictors of duration of abiraterone acetate in men with castration-resistant prostate cancer.Prostate Cancer Prostatic Dis. 2016 Dec;19(4):398-405. doi: 10.1038/pcan.2016.31. Epub 2016 Aug 9. Prostate Cancer Prostatic Dis. 2016. PMID: 27502737 Free PMC article.
-
Management of metastatic castration-resistant prostate cancer: Insights from urology experts in Thailand.Prostate Int. 2017 Mar;5(1):1-7. doi: 10.1016/j.prnil.2017.01.002. Epub 2017 Jan 11. Prostate Int. 2017. PMID: 28352616 Free PMC article. Review.
-
Efficacy and Safety of Abiraterone Acetate and Enzalutamide for the Treatment of Metastatic Castration-Resistant Prostate Cancer: A Systematic Review and Meta-Analysis.Front Oncol. 2021 Aug 27;11:732599. doi: 10.3389/fonc.2021.732599. eCollection 2021. Front Oncol. 2021. PMID: 34513709 Free PMC article.
References
-
- Lotan TL, Epstein JI. Clinical implications of changing definitions within the Gleason grading system. Nat Rev Urol 2010; 7: 136–142. - PubMed
-
- Epstein JI, Allsbrook WC Jr, Amin MB, Egevad LL. The 2005 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma. Am J Surg Pathol 2005; 29: 1228–1242. - PubMed
-
- Dong F, Wang C, Farris AB et al. . Impact on the clinical outcome of prostate cancer by the 2005 International Society of Urological Pathology modified Gleason grading system. Am J Surg Pathol 2012; 36: 838–843. - PubMed
-
- Freedland SJ, Humphreys EB, Mangold LA et al. . Risk of prostate cancer-specific mortality following biochemical recurrence after radical prostatectomy. JAMA 2005; 294: 433–439. - PubMed
-
- Armstrong AJ, Tannock IF, de Wit R et al. . The development of risk groups in men with metastatic castration-resistant prostate cancer based on risk factors for PSA decline and survival. Eur J Cancer 2010; 46: 517–525. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

