The risk of major cardiac malformations associated with paroxetine use during the first trimester of pregnancy: a systematic review and meta-analysis
- PMID: 26613360
- PMCID: PMC4799922
- DOI: 10.1111/bcp.12849
The risk of major cardiac malformations associated with paroxetine use during the first trimester of pregnancy: a systematic review and meta-analysis
Abstract
Aims: The aim of this study was to perform an up-to-date meta-analysis on the risk of cardiac malformations associated with gestational exposure to paroxetine, taking into account indication, study design and reference category.
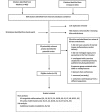
Method: A systematic review of studies published between 1966 and November 2015 was conducted using embase and MEDLINE. Studies reporting major malformations with first trimester exposure to paroxetine were included. Potentially relevant articles were assessed and relevant data extracted to calculate risk estimates. Outcomes included any major malformations and major cardiac malformations. Pooled odds ratios and 95% confidence intervals were calculated using random-effects models.
Results: Twenty-three studies were included. Compared with non-exposure to paroxetine, first trimester use of paroxetine was associated with an increased risk of any major congenital malformations combined (pooled OR 1.23, 95% CI 1.10, 1.38; n = 15 studies), major cardiac malformations (pooled OR 1.28, 95% CI 1.11, 1.47; n = 18 studies), specifically bulbus cordis anomalies and anomalies of cardiac septal closure (pooled OR 1.42, 95% CI 1.07, 1.89; n = 8 studies), atrial septal defects (pooled OR 2.38, 95% CI 1.14, 4.97; n = 4 studies) and right ventricular outflow track defect (pooled OR 2.29, 95% CI 1.06, 4.93; n = 4 studies). Although the estimates varied depending on the comparator group, study design and malformation detection period, a trend towards increased risk was observed.
Conclusions: Paroxetine use during the first trimester of pregnancy is associated with an increased risk of any major congenital malformations and cardiac malformations. The increase in risk is not dependent on the study method or population.
Keywords: cardiac malformations; major malformations; meta-analysis; paroxetine; pregnancy.
© 2015 The British Pharmacological Society.
Figures

Comment in
-
Meta-analysis requires independent observations and freedom from bias.Br J Clin Pharmacol. 2016 Jun;81(6):1191-3. doi: 10.1111/bcp.12898. Epub 2016 Mar 23. Br J Clin Pharmacol. 2016. PMID: 26845509 Free PMC article. No abstract available.
-
Paroxetine use during pregnancy and the risk of cardiac defects.Br J Clin Pharmacol. 2016 Aug;82(2):566-7. doi: 10.1111/bcp.12979. Epub 2016 May 20. Br J Clin Pharmacol. 2016. PMID: 27197980 Free PMC article. No abstract available.
References
-
- Marcus SM. Depression during pregnancy: rates, risks and consequences–motherisk update 2008. Can J Clin Pharmacol 2009; 16: e15–22. - PubMed
-
- Grigoriadis S, Robinson GE. Gender issues in depression. Ann Clin Psychiatry 2007; 19: 247–55. - PubMed
-
- Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age‐of‐onset distributions of DSM‐IV disorders in the national comorbidity survey replication. Arch Gen Psychiatry 2005; 62: 593–602. - PubMed
-
- Bennett HA, Einarson A, Taddio A, Koren G, Einarson TR. Prevalence of depression during pregnancy: systematic review. Obstet Gynecol 2004; 103: 698–709. - PubMed
-
- Silva R, Jansen K, Souza L, Quevedo L, Barbosa L, Moraes I, Horta B, Pinheiro R. Sociodemographic risk factors of perinatal depression: a cohort study in the public health care system. Rev Bras Psiquiatr 2012; 34: 143–8. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

