HIV pre-exposure prophylaxis in transgender women: a subgroup analysis of the iPrEx trial
- PMID: 26614965
- PMCID: PMC5111857
- DOI: 10.1016/S2352-3018(15)00206-4
HIV pre-exposure prophylaxis in transgender women: a subgroup analysis of the iPrEx trial
Abstract
Background: Pre-exposure prophylaxis (PrEP) with oral emtricitabine and tenofovir disoproxil fumarate is used to prevent the sexual acquisition of HIV in groups at high risk such as transgender women. We used data from the iPrEx study to assess PrEP efficacy, effectiveness, and adherence in transgender women.
Methods: The iPrEx trial was a randomised controlled trial of PrEP with oral emtricitabine plus tenofovir disoproxil fumarate compared with placebo in men who have sex with men (MSM) and transgender women, followed by an open-label extension. Drug concentrations were measured in blood by liquid chromatography and tandem mass spectroscopy. We did unplanned exploratory analyses to investigate differences in PrEP outcomes among transgender women and between transgender women and MSM.
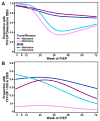
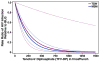
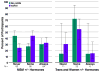
Findings: Of the 2499 participants enrolled in the randomised controlled trial, 29 (1%) identified as women, 296 (12%) identified as trans, 14 (1%) identified as men but reported use of feminising hormones, such that 339 (14%) reported one or more characteristics and are classified as transgender women for the purpose of this study. Compared with MSM, transgender women more frequently reported transactional sex, receptive anal intercourse without a condom, or more than five partners in the past 3 months. Among transgender women, there were 11 HIV infections in the PrEP group and ten in the placebo group (hazard ratio 1·1, 95% CI 0·5-2·7). In the PrEP group, drug was detected in none of the transgender women at the seroconversion visit, six (18%) of 33 seronegative transgender women (p=0·31), and 58 (52%) of 111 seronegative MSM (p<0·0001). PrEP use was not linked to behavioural indicators of HIV risk among transgender women, whereas MSM at highest risk were more adherent.
Interpretation: PrEP seems to be effective in preventing HIV acquisition in transgender women when taken, but there seem to be barriers to adherence, particularly among those at the most risk. Studies of PrEP use in transgender women populations should be designed and tailored specifically for this population, rather than adapted from or subsumed into studies of MSM.
Funding: US National Institutes of Health and the Bill & Melinda Gates Foundation.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures
Comment in
-
Uptake and effectiveness of PrEP for transgender women.Lancet HIV. 2015 Dec;2(12):e502-3. doi: 10.1016/S2352-3018(15)00224-6. Epub 2015 Nov 6. Lancet HIV. 2015. PMID: 26614960 Free PMC article. No abstract available.
-
PrEP adherence among trans women in Brazil-access needed for this key population.Lancet HIV. 2016 May;3(5):e200. doi: 10.1016/S2352-3018(16)30022-4. Lancet HIV. 2016. PMID: 27126485 No abstract available.
References
-
- Kuba S. Sexually and gender-variant individuals: International and multicultural perspectives. In: Lowman RL, editor. Internationalizing multiculturalism : expanding professional competencies in a globalized world. 1. Washington, D.C: American Psychological Association; 2013. pp. 109–42.
-
- Deutsch MB, Keatley J, Sevelius J, Shade SB. Collection of gender identity data using electronic medical records: survey of current end-user practices. J Assoc Nurses AIDS Care. 2014 Nov-Dec;25(6):657–63. - PubMed
-
- Cahill S, Makadon H. Sexual Orientation and Gender Identity Data Collection Update: U.S. Government Takes Steps to Promote Sexual Orientation and Gender Identity Data Collection Through Meaningful Use Guidelines. LGBT Health. 2014;1(3):157–60. - PubMed
-
- Baral SD, Poteat T, Stromdahl S, Wirtz AL, Guadamuz TE, Beyrer C. Worldwide burden of HIV in transgender women: a systematic review and meta-analysis. Lancet Infect Dis. 2013 Mar;13(3):214–22. - PubMed

