High-Intensity Focused Ultrasound: Current Status for Image-Guided Therapy
- PMID: 26622104
- PMCID: PMC4640913
- DOI: 10.1055/s-0035-1564793
High-Intensity Focused Ultrasound: Current Status for Image-Guided Therapy
Abstract
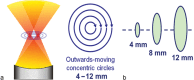
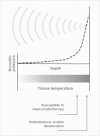
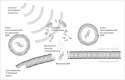
Image-guided high-intensity focused ultrasound (HIFU) is an innovative therapeutic technology, permitting extracorporeal or endocavitary delivery of targeted thermal ablation while minimizing injury to the surrounding structures. While ultrasound-guided HIFU was the original image-guided system, MR-guided HIFU has many inherent advantages, including superior depiction of anatomic detail and superb real-time thermometry during thermoablation sessions, and it has recently demonstrated promising results in the treatment of both benign and malignant tumors. HIFU has been employed in the management of prostate cancer, hepatocellular carcinoma, uterine leiomyomas, and breast tumors, and has been associated with success in limited studies for palliative pain management in pancreatic cancer and bone tumors. Nonthermal HIFU bioeffects, including immune system modulation and targeted drug/gene therapy, are currently being explored in the preclinical realm, with an emphasis on leveraging these therapeutic effects in the care of the oncology patient. Although still in its early stages, the wide spectrum of therapeutic capabilities of HIFU offers great potential in the field of image-guided oncologic therapy.
Keywords: HIFU; ablation; focused ultrasound surgery; image guided; interventional oncology; interventional radiology.
Figures








References
-
- Wood R W, Loomis A L. The physical and biological effects of high frequency sound waves of great intensity. Philos Mag and J of Science. 1927;4(22):417–436.
-
- Fry W J, Barnard J W, Fry F J, Brennan J F. Ultrasonically produced localized selective lesions in the central nervous system. Am J Phys Med. 1955;34(3):413–423. - PubMed
-
- Susani M, Madersbacher S, Kratzik C, Vingers L, Marberger M. Morphology of tissue destruction induced by focused ultrasound. Eur Urol. 1993;23 01:34–38. - PubMed
-
- Madersbacher S, Pedevilla M, Vingers L, Susani M, Marberger M. Effect of high-intensity focused ultrasound on human prostate cancer in vivo. Cancer Res. 1995;55(15):3346–3351. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

