Shifting eating to the circadian rest phase misaligns the peripheral clocks with the master SCN clock and leads to a metabolic syndrome
- PMID: 26627260
- PMCID: PMC4672793
- DOI: 10.1073/pnas.1519807112
Shifting eating to the circadian rest phase misaligns the peripheral clocks with the master SCN clock and leads to a metabolic syndrome
Abstract
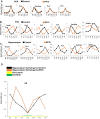
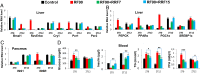
The light-entrained master central circadian clock (CC) located in the suprachiasmatic nucleus (SCN) not only controls the diurnal alternance of the active phase (the light period of the human light-dark cycle, but the mouse dark period) and the rest phase (the human dark period, but the mouse light period), but also synchronizes the ubiquitous peripheral CCs (PCCs) with these phases to maintain homeostasis. We recently elucidated in mice the molecular signals through which metabolic alterations induced on an unusual feeding schedule, taking place during the rest phase [i.e., restricted feeding (RF)], creates a 12-h PCC shift. Importantly, a previous study showed that the SCN CC is unaltered during RF, which creates a misalignment between the RF-shifted PCCs and the SCN CC-controlled phases of activity and rest. However, the molecular basis of SCN CC insensitivity to RF and its possible pathological consequences are mostly unknown. Here we deciphered, at the molecular level, how RF creates this misalignment. We demonstrate that the PPARα and glucagon receptors, the two instrumental transducers in the RF-induced shift of PCCs, are not expressed in the SCN, thereby preventing on RF a shift of the master SCN CC and creating the misalignment. Most importantly, this RF-induced misalignment leads to a misexpression (with respect to their normal physiological phase of expression) of numerous CC-controlled homeostatic genes, which in the long term generates in RF mice a number of metabolic pathologies including diabetes, obesity, and metabolic syndrome, which have been reported in humans engaged in shift work schedules.
Keywords: circadian clocks misalignment; diabetes; metabolic syndrome; mouse; shift work.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







Similar articles
-
Shifting the feeding of mice to the rest phase creates metabolic alterations, which, on their own, shift the peripheral circadian clocks by 12 hours.Proc Natl Acad Sci U S A. 2015 Dec 1;112(48):E6683-90. doi: 10.1073/pnas.1519735112. Epub 2015 Nov 16. Proc Natl Acad Sci U S A. 2015. PMID: 26627259 Free PMC article.
-
Feeding and adrenal entrainment stimuli are both necessary for normal circadian oscillation of peripheral clocks in mice housed under different photoperiods.Chronobiol Int. 2015 Mar;32(2):195-210. doi: 10.3109/07420528.2014.962655. Epub 2014 Oct 6. Chronobiol Int. 2015. PMID: 25286135
-
The circadian clock controls fluctuations of colonic cell proliferation during the light/dark cycle via feeding behavior in mice.Chronobiol Int. 2015;32(8):1145-55. doi: 10.3109/07420528.2015.1065415. Epub 2015 Sep 16. Chronobiol Int. 2015. PMID: 26376208
-
Misalignment of Circadian Rhythms in Diet-Induced Obesity.Adv Exp Med Biol. 2024;1460:27-71. doi: 10.1007/978-3-031-63657-8_2. Adv Exp Med Biol. 2024. PMID: 39287848 Review.
-
Circadian Rhythms in Diet-Induced Obesity.Adv Exp Med Biol. 2017;960:19-52. doi: 10.1007/978-3-319-48382-5_2. Adv Exp Med Biol. 2017. PMID: 28585194 Review.
Cited by
-
Hypothalamic REV-ERB nuclear receptors control diurnal food intake and leptin sensitivity in diet-induced obese mice.J Clin Invest. 2021 Jan 4;131(1):e140424. doi: 10.1172/JCI140424. J Clin Invest. 2021. PMID: 33021965 Free PMC article.
-
Emerging role of circadian clock disruption in alcohol-induced liver disease.Am J Physiol Gastrointest Liver Physiol. 2018 Sep 1;315(3):G364-G373. doi: 10.1152/ajpgi.00010.2018. Epub 2018 May 31. Am J Physiol Gastrointest Liver Physiol. 2018. PMID: 29848023 Free PMC article. Review.
-
Plasma Cell-Free DNA Methylomics of Bipolar Disorder With and Without Rapid Cycling.Front Neurosci. 2021 Nov 30;15:774037. doi: 10.3389/fnins.2021.774037. eCollection 2021. Front Neurosci. 2021. PMID: 34916903 Free PMC article.
-
Nutrigenetics and Nutrimiromics of the Circadian System: The Time for Human Health.Int J Mol Sci. 2016 Feb 26;17(3):299. doi: 10.3390/ijms17030299. Int J Mol Sci. 2016. PMID: 26927084 Free PMC article. Review.
-
Timing of meals: when is as critical as what and how much.Am J Physiol Endocrinol Metab. 2017 May 1;312(5):E369-E380. doi: 10.1152/ajpendo.00295.2016. Epub 2017 Jan 31. Am J Physiol Endocrinol Metab. 2017. PMID: 28143856 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

