Cardiac contraction activates endocardial Notch signaling to modulate chamber maturation in zebrafish
- PMID: 26628092
- PMCID: PMC4712836
- DOI: 10.1242/dev.125724
Cardiac contraction activates endocardial Notch signaling to modulate chamber maturation in zebrafish
Abstract
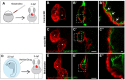
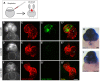
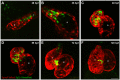
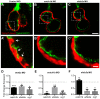
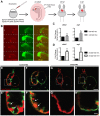
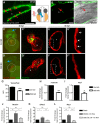
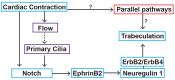
Congenital heart disease often features structural abnormalities that emerge during development. Accumulating evidence indicates a crucial role for cardiac contraction and the resulting fluid forces in shaping the heart, yet the molecular basis of this function is largely unknown. Using the zebrafish as a model of early heart development, we investigated the role of cardiac contraction in chamber maturation, focusing on the formation of muscular protrusions called trabeculae. By genetic and pharmacological ablation of cardiac contraction, we showed that cardiac contraction is required for trabeculation through its role in regulating notch1b transcription in the ventricular endocardium. We also showed that Notch1 activation induces expression of ephrin b2a (efnb2a) and neuregulin 1 (nrg1) in the endocardium to promote trabeculation and that forced Notch activation in the absence of cardiac contraction rescues efnb2a and nrg1 expression. Using in vitro and in vivo systems, we showed that primary cilia are important mediators of fluid flow to stimulate Notch expression. Together, our findings describe an essential role for cardiac contraction-responsive transcriptional changes in endocardial cells to regulate cardiac chamber maturation.
Keywords: Blood flow; Cardiac chamber maturation; Cardiac contraction; Neuregulin 1; Notch; Primary cilia; Trabeculation; Zebrafish.
© 2015. Published by The Company of Biologists Ltd.
Conflict of interest statement
The authors declare no competing or financial interests.
Figures
References
-
- Anderson K. V. (2006). Cilia and Hedgehog signaling in the mouse embryo. Harvey Lect. 102, 103-115. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

