c-Met as a Target for Personalized Therapy
- PMID: 26628860
- PMCID: PMC4659440
- DOI: 10.4137/TOG.S30534
c-Met as a Target for Personalized Therapy
Abstract
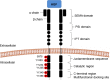
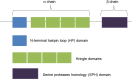
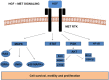
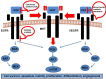
MET and its ligand HGF are involved in many biological processes, both physiological and pathological, making this signaling pathway an attractive therapeutic target in oncology. Downstream signaling effects are transmitted via mitogen-activated protein kinase (MAPK), PI3K (phosphoinositide 3-kinase protein kinase B)/AKT, signal transducer and activator of transcription proteins (STAT), and nuclear factor-κB. The final output of the terminal effector components of these pathways is activation of cytoplasmic and nuclear processes leading to increases in cell proliferation, survival, mobilization and invasive capacity. In addition to its role as an oncogenic driver, increasing evidence implicates MET as a common mechanism of resistance to targeted therapies including EGFR and VEGFR inhibitors. In the present review, we summarize the current knowledge on the role of the HGF-MET signaling pathway in cancer and its therapeutic targeting (HGF activation inhibitors, HGF inhibitors, MET antagonists and selective/nonselective MET kinase inhibitors). Recent advances in understanding the role of this pathway in the resistance to current anticancer strategies used in lung, kidney and pancreatic cancer are discussed.
Keywords: HGF; MET; NSCLC; drug resistance; pancreatic cancer; renal cancer; targeted therapy.
Figures

References
-
- Peters S, Adjei AA. MET: a promising anticancer therapeutic target. Nat Rev Clin Oncol. 2012;9(6):314–26. - PubMed
-
- Maroun CR, Rowlands T. The Met receptor tyrosine kinase: a key player in oncogenesis and drug resistance. Pharmacol Ther. 2014;142(3):316–38. - PubMed
-
- Cooper CS, Park M, Blair DG, et al. Molecular cloning of a new transforming gene from a chemically transformed human cell line. Nature. 1984;311(5981):29–33. - PubMed
-
- Gherardi E, Birchmeier W, Birchmeier C, et al. Targeting MET in cancer: rationale and progress. Nat Rev Cancer. 2012;12(2):89–103. - PubMed
-
- Peschard P, Fornier TM, Lamorte L, et al. Mutation of the c-Cbl TKB domain binding site on the Met receptor tyrosine kinase converts it into a transforming protein. Mol Cell. 2001;8(5):995–1004. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
