The styrene-maleic acid copolymer: a versatile tool in membrane research
- PMID: 26639665
- PMCID: PMC4698303
- DOI: 10.1007/s00249-015-1093-y
The styrene-maleic acid copolymer: a versatile tool in membrane research
Abstract
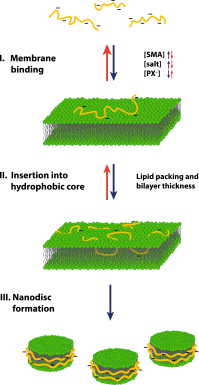
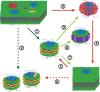
A new and promising tool in membrane research is the detergent-free solubilization of membrane proteins by styrene-maleic acid copolymers (SMAs). These amphipathic molecules are able to solubilize lipid bilayers in the form of nanodiscs that are bounded by the polymer. Thus, membrane proteins can be directly extracted from cells in a water-soluble form while conserving a patch of native membrane around them. In this review article, we briefly discuss current methods of membrane protein solubilization and stabilization. We then zoom in on SMAs, describe their physico-chemical properties, and discuss their membrane-solubilizing effect. This is followed by an overview of studies in which SMA has been used to isolate and investigate membrane proteins. Finally, potential future applications of the methodology are discussed for structural and functional studies on membrane proteins in a near-native environment and for characterizing protein-lipid and protein-protein interactions.
Keywords: Lipid–protein interactions; Lipodisq; Membrane proteins; Native nanodiscs; SMALPs; Styrene–maleic acid copolymers.
Figures

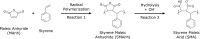



References
-
- Alfrey T, Lavin E. The copolymerization of styrene and maleic anhydride. J Am Chem Soc. 1945;67:2044–2045. doi: 10.1021/ja01227a502. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

