Breakdown of interlocking domains may contribute to formation of membranous globules and lens opacity in ephrin-A5(-/-) mice
- PMID: 26643403
- PMCID: PMC4842153
- DOI: 10.1016/j.exer.2015.11.017
Breakdown of interlocking domains may contribute to formation of membranous globules and lens opacity in ephrin-A5(-/-) mice
Abstract
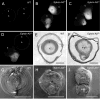
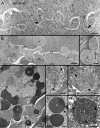
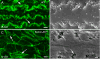
Ephrin-A5, a ligand of the Eph family of receptor tyrosine kinases, plays a key role in lens fiber cell packing and cell-cell adhesion, with approximately 87% of ephrin-A5(-/-) mice develop nuclear cataracts. Here, we investigated the extensive formation of light-scattering globules associated with breakdown of interlocking protrusions during lens opacification in ephrin-A5(-/-) mice. Lenses from wild-type (WT) and ephrin-A5(-/-) mice between 2 and 21 weeks old were studied with light and electron microscopy, immunofluorescence labeling, freeze-fracture TEM and filipin cytochemistry for membrane cholesterol detection. Lens opacities with various densities were first observed in ephrin-A5(-/-) mice at around 60 days old. Dense cataracts in the mutant lenses were seen primarily in the nuclear region surrounded by transparent cortices from all eyes examined. We confirmed that a majority of nuclear cataracts were dislocated posteriorly and ruptured the thinner posterior lens capsule. SEM analysis indicated that numerous interlocking protrusions and wavy ridge-and-valley membrane surfaces in deep cortical and nuclear fibers did not cause lens opacity in both transparent ephrin-A5(-/-) and WT mice. In contrast, abundant isolated membranous globules of approximately 1000 nm in size were distributed randomly along the intact fiber cells during early stage of all ephrin-A5(-/-) cataracts examined. A further examination using both SEM and TEM revealed that isolated globules were generated from the disintegrated interlocking protrusions originally located along the corners of hexagonal fiber cells. Freeze-fracture TEM further revealed the association of square-array aquaporin junctions with both isolated globules and interlocking membrane domains. This study reports for the first time that disrupted interlocking protrusions are the source of numerous large membranous globules that contribute to light scattering and nuclear cataracts in the ephrin-A5(-/-) mice. Our results further suggest that dissociations of N-cadherin and adherens junctions in the associated interlocking domains may result in the formation of isolated globules and nuclear opacities in the ephrin-A5(-/-) mice.
Keywords: Adherens junction; Ephrin-A5 knockout mouse; Interlocking domain; Lens; Membranous globule; N-cadherin; Nuclear cataract.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures






References
-
- Atreya PL, Barnes J, Katar M, Alcala J, Maisel H. N-cadherin of the human lens. Curr Eye Res. 1989;8:947–956. - PubMed
-
- Bagchi M, Katar M, Lewis J, Maisel H. Associated proteins of lens adherens junction. J Cell Biochem. 2002;86:700–703. - PubMed
-
- Bhatnagar A, Ansari NH, Wang L, Khanna P, Wang C, Srivastava SK. Calcium-mediated disintegrative globulization of isolated ocular lens fibers mimics cataractogenesis. Exp Eye Res. 1995;61:303–310. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

