Proteome Profiling of the Rhodobacter capsulatus Molybdenum Response Reveals a Role of IscN in Nitrogen Fixation by Fe-Nitrogenase
- PMID: 26644433
- PMCID: PMC4751811
- DOI: 10.1128/JB.00750-15
Proteome Profiling of the Rhodobacter capsulatus Molybdenum Response Reveals a Role of IscN in Nitrogen Fixation by Fe-Nitrogenase
Abstract
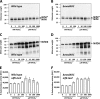
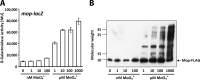
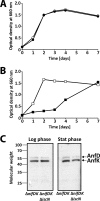
Rhodobacter capsulatus is capable of synthesizing two nitrogenases, a molybdenum-dependent nitrogenase and an alternative Mo-free iron-only nitrogenase, enabling this diazotroph to grow with molecular dinitrogen (N2) as the sole nitrogen source. Here, the Mo responses of the wild type and of a mutant lacking ModABC, the high-affinity molybdate transporter, were examined by proteome profiling, Western analysis, epitope tagging, and lacZ reporter fusions. Many Mo-controlled proteins identified in this study have documented or presumed roles in nitrogen fixation, demonstrating the relevance of Mo control in this highly ATP-demanding process. The levels of Mo-nitrogenase, NifHDK, and the Mo storage protein, Mop, increased with increasing Mo concentrations. In contrast, Fe-nitrogenase, AnfHDGK, and ModABC, the Mo transporter, were expressed only under Mo-limiting conditions. IscN was identified as a novel Mo-repressed protein. Mo control of Mop, AnfHDGK, and ModABC corresponded to transcriptional regulation of their genes by the Mo-responsive regulators MopA and MopB. Mo control of NifHDK and IscN appeared to be more complex, involving different posttranscriptional mechanisms. In line with the simultaneous control of IscN and Fe-nitrogenase by Mo, IscN was found to be important for Fe-nitrogenase-dependent diazotrophic growth. The possible role of IscN as an A-type carrier providing Fe-nitrogenase with Fe-S clusters is discussed.
Importance: Biological nitrogen fixation is a central process in the global nitrogen cycle by which the abundant but chemically inert dinitrogen (N2) is reduced to ammonia (NH3), a bioavailable form of nitrogen. Nitrogen reduction is catalyzed by nitrogenases found in diazotrophic bacteria and archaea but not in eukaryotes. All diazotrophs synthesize molybdenum-dependent nitrogenases. In addition, some diazotrophs, including Rhodobacter capsulatus, possess catalytically less efficient alternative Mo-free nitrogenases, whose expression is repressed by Mo. Despite the importance of Mo in biological nitrogen fixation, this is the first study analyzing the proteome-wide Mo response in a diazotroph. IscN was recognized as a novel member of the molybdoproteome in R. capsulatus. It was dispensable for Mo-nitrogenase activity but supported diazotrophic growth under Mo-limiting conditions.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

