Targeting Transcription Factors in Cancer
- PMID: 26645049
- PMCID: PMC4669894
- DOI: 10.1016/j.trecan.2015.07.001
Targeting Transcription Factors in Cancer
Abstract
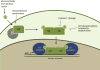
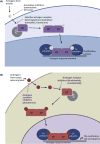
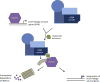
Transcription factors (TFs) are commonly deregulated in the pathogenesis of human cancer and are a major class of cancer cell dependencies. Consequently, targeting of TFs can be highly effective in treating particular malignancies, as highlighted by the clinical efficacy of agents that target nuclear hormone receptors. In this review we discuss recent advances in our understanding of TFs as drug targets in oncology, with an emphasis on the emerging chemical approaches to modulate TF function. The remarkable diversity and potency of TFs as drivers of cell transformation justifies a continued pursuit of TFs as therapeutic targets for drug discovery.
Figures


References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
