Characterization of a Novel Human-Specific STING Agonist that Elicits Antiviral Activity Against Emerging Alphaviruses
- PMID: 26646986
- PMCID: PMC4672893
- DOI: 10.1371/journal.ppat.1005324
Characterization of a Novel Human-Specific STING Agonist that Elicits Antiviral Activity Against Emerging Alphaviruses
Abstract
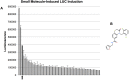
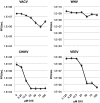
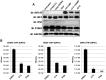
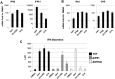
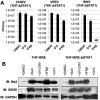
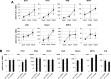
Pharmacologic stimulation of innate immune processes represents an attractive strategy to achieve multiple therapeutic outcomes including inhibition of virus replication, boosting antitumor immunity, and enhancing vaccine immunogenicity. In light of this we sought to identify small molecules capable of activating the type I interferon (IFN) response by way of the transcription factor IFN regulatory factor 3 (IRF3). A high throughput in vitro screen yielded 4-(2-chloro-6-fluorobenzyl)-N-(furan-2-ylmethyl)-3-oxo-3,4-dihydro-2H-benzo[b][1,4]thiazine-6-carboxamide (referred to herein as G10), which was found to trigger IRF3/IFN-associated transcription in human fibroblasts. Further examination of the cellular response to this molecule revealed expression of multiple IRF3-dependent antiviral effector genes as well as type I and III IFN subtypes. This led to the establishment of a cellular state that prevented replication of emerging Alphavirus species including Chikungunya virus, Venezuelan Equine Encephalitis virus, and Sindbis virus. To define cellular proteins essential to elicitation of the antiviral activity by the compound we employed a reverse genetics approach that utilized genome editing via CRISPR/Cas9 technology. This allowed the identification of IRF3, the IRF3-activating adaptor molecule STING, and the IFN-associated transcription factor STAT1 as required for observed gene induction and antiviral effects. Biochemical analysis indicates that G10 does not bind to STING directly, however. Thus the compound may represent the first synthetic small molecule characterized as an indirect activator of human STING-dependent phenotypes. In vivo stimulation of STING-dependent activity by an unrelated small molecule in a mouse model of Chikungunya virus infection blocked viremia demonstrating that pharmacologic activation of this signaling pathway may represent a feasible strategy for combating emerging Alphaviruses.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

