Assessment of the Therapeutic Potential of Metallothionein-II Application in Focal Cerebral Ischemia In Vitro and In Vivo
- PMID: 26658636
- PMCID: PMC4682799
- DOI: 10.1371/journal.pone.0144035
Assessment of the Therapeutic Potential of Metallothionein-II Application in Focal Cerebral Ischemia In Vitro and In Vivo
Abstract
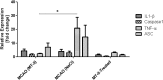
Metallothionein-II (MT-II) is an ubiquitously expressed small-molecular-weight protein and highly induced in various species and tissues upon stress, inflammation, and ischemia. MT-deficiency exacerbates ischemic injury in rodent stroke models in vitro and in vivo. However, there is conflicting data on the potential neuroprotective effect of exogenously applied metallothionein. Thus, we applied MT-II in an in vitro stroke model and intraperitoneally (i.p.) in two in vivo standard models of transient middle cerebral artery occlusion (MCAO) (a 'stringent' one [60 min MCAO/48 h reperfusion] and a 'mild' one [30 min MCAO/72 h reperfusion]), as well as i.v. together with recombinant tissue plasminogen activator (rtPA) to evaluate if exogenous MT-II-application protects against ischemic stroke. Whereas MT-II did not protect against 60 min MCAO, there was a significant reduction of direct and indirect infarct volumes and neurological deficit in the MT-II (i.p.) treated animals in the 'mild' model at 3d after MCAO. Furthermore, MT-II also improved survival of the mice after MCAO, suppressed TNF-α mRNA induction in ischemic brain tissue, and protected primary neuronal cells against oxygen-glucose-deprivation in vitro. Thus, exogenous application of MT-II protects against ischemic injury in vitro and in vivo. However, long-term studies with different species and larger sampling sizes are required before a clinical use can be envisaged.
Conflict of interest statement
Figures




Similar articles
-
Neuroprotective effect of erythropoietin, and role of metallothionein-1 and -2, in permanent focal cerebral ischemia.Neuroscience. 2007 Aug 10;148(1):105-14. doi: 10.1016/j.neuroscience.2007.04.063. Epub 2007 Jul 12. Neuroscience. 2007. PMID: 17624681
-
Ruscogenin reduces cerebral ischemic injury via NF-κB-mediated inflammatory pathway in the mouse model of experimental stroke.Eur J Pharmacol. 2013 Aug 15;714(1-3):303-11. doi: 10.1016/j.ejphar.2013.07.036. Epub 2013 Jul 30. Eur J Pharmacol. 2013. PMID: 23911884
-
Protective effects of PJ34, a novel, potent inhibitor of poly(ADP-ribose) polymerase (PARP) in in vitro and in vivo models of stroke.Int J Mol Med. 2001 Mar;7(3):255-60. Int J Mol Med. 2001. PMID: 11179503
-
Role of Metallothionein-1 and Metallothionein-2 in the Neuroprotective Mechanism of Sevoflurane Preconditioning in Mice.J Mol Neurosci. 2020 May;70(5):713-723. doi: 10.1007/s12031-020-01481-3. Epub 2020 Jan 17. J Mol Neurosci. 2020. PMID: 31953609
-
Protective role of metallothioneins in the injured mammalian brain.Rev Neurosci. 2004;15(3):157-66. doi: 10.1515/revneuro.2004.15.3.157. Rev Neurosci. 2004. PMID: 15357139 Review.
Cited by
-
RNA Sequencing Analyses Reveal the Potential Anti-Inflammatory Mechanisms of Acacetin Against ODG/R Injuries in Microglia.J Inflamm Res. 2024 Jun 5;17:3641-3652. doi: 10.2147/JIR.S465093. eCollection 2024. J Inflamm Res. 2024. PMID: 38855167 Free PMC article.
-
Metallothionein in Brain Disorders.Oxid Med Cell Longev. 2017;2017:5828056. doi: 10.1155/2017/5828056. Epub 2017 Sep 20. Oxid Med Cell Longev. 2017. PMID: 29085556 Free PMC article. Review.
-
Focusing on the protective effects of metallothionein-I/II in cerebral ischemia.Neural Regen Res. 2016 May;11(5):721-2. doi: 10.4103/1673-5374.182689. Neural Regen Res. 2016. PMID: 27335546 Free PMC article. No abstract available.
-
Influence of Transgenic Metallothionein-1 on Gliosis, CA1 Neuronal Loss, and Brain Metal Levels of the Tg2576 Mouse Model of Alzheimer's Disease.Int J Mol Sci. 2017 Jan 26;18(2):251. doi: 10.3390/ijms18020251. Int J Mol Sci. 2017. PMID: 28134760 Free PMC article.
-
Metallothionein I as a direct link between therapeutic hematopoietic stem/progenitor cells and cerebral protection in stroke.FASEB J. 2018 May;32(5):2381-2394. doi: 10.1096/fj.201700746R. Epub 2017 Dec 21. FASEB J. 2018. PMID: 29269399 Free PMC article.
References
-
- Feigin VL, Forouzanfar MH, Krishnamurthi R, Mensah GA, Connor M, Bennett DA, et al. Global Burden of Diseases, Injuries, and Risk Factors Study 2010 (GBD 2010) and the GBD Stroke Experts Group (2014). Global and regional burden of stroke during 1990–2010: findings from the Global Burden of Disease Study 2010. Lancet. 2014; 383: 245–54. - PMC - PubMed
-
- Dirnagl U, Iadecola C, Moskowitz MA. Pathobiology of ischaemic stroke: an integrated view. Trends Neurosci. 1999; 22: 391–397. - PubMed
-
- O'Collins VE, Macleod MR, Donnan GA, Horky LL, van der Worp BH, Howells DW. 1,026 experimental treatments in acute stroke. Ann Neurol. 2006; 59: 467–77. - PubMed
-
- Chung RS, Hidalgo J, West AK. New insight into the molecular pathways of metallothionein-mediated neuroprotection and regeneration. J Neurochem. 2008; 104: 14–20. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

