Neuronal NCX1 overexpression induces stroke resistance while knockout induces vulnerability via Akt
- PMID: 26661211
- PMCID: PMC5076784
- DOI: 10.1177/0271678X15611913
Neuronal NCX1 overexpression induces stroke resistance while knockout induces vulnerability via Akt
Abstract
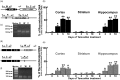
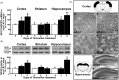
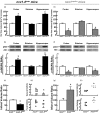
Three different Na+/Ca2+ exchanger (NCX) isoforms, NCX1, NCX2, and NCX3, are expressed in brain where they play a relevant role in maintaining Na+ and Ca2+ homeostasis. Although the neuroprotective roles of NCX2 and NCX3 in stroke have been elucidated, the relevance of NCX1 is still unknown because of embryonic lethality of its knocking-out, heart dysfunctions when it is overexpressed, and the lack of selectivity in currently available drugs. To overcome these limitations we generated two conditional genetically modified mice that upon tamoxifen administration showed a selective decrease or increase of NCX1 in cortical and hippocampal neurons. Interestingly, in cortex and hippocampus NCX1 overexpression increased, where NCX1 knock-out reduced, both exchanger activity and Akt1 phosphorylation, a neuronal survival signaling. More important, mice overexpressing NCX1 showed a reduced ischemic volume and an amelioration of focal and general deficits when subjected to transient middle cerebral artery occlusion. Conversely, NCX1-knock-out mice displayed a worsening of brain damage, focal and neurological deficits with a decrease in Akt phosphorylation. These results support the idea that NCX1 overexpression/activation may represent a feasible therapeutic opportunity in stroke intervention.
Keywords: Akt; Cre-LoxP; NCX1; Na+-Ca2+ exchanger; stroke.
© The Author(s) 2015.
Figures



Similar articles
-
Involvement of the Na+/Ca2+ exchanger isoform 1 (NCX1) in neuronal growth factor (NGF)-induced neuronal differentiation through Ca2+-dependent Akt phosphorylation.J Biol Chem. 2015 Jan 16;290(3):1319-31. doi: 10.1074/jbc.M114.555516. Epub 2014 Nov 21. J Biol Chem. 2015. PMID: 25416782 Free PMC article.
-
Genetically modified mice as a strategy to unravel the role played by the Na(+)/Ca (2+) exchanger in brain ischemia and in spatial learning and memory deficits.Adv Exp Med Biol. 2013;961:213-22. doi: 10.1007/978-1-4614-4756-6_18. Adv Exp Med Biol. 2013. PMID: 23224882 Review.
-
Preferential involvement of Na⁺/Ca²⁺ exchanger type-1 in the brain damage caused by transient focal cerebral ischemia in mice.Biochem Biophys Res Commun. 2012 Dec 14;429(3-4):186-90. doi: 10.1016/j.bbrc.2012.10.114. Epub 2012 Nov 5. Biochem Biophys Res Commun. 2012. PMID: 23137542
-
The two isoforms of the Na+/Ca2+ exchanger, NCX1 and NCX3, constitute novel additional targets for the prosurvival action of Akt/protein kinase B pathway.Mol Pharmacol. 2008 Mar;73(3):727-37. doi: 10.1124/mol.107.042549. Epub 2007 Dec 13. Mol Pharmacol. 2008. PMID: 18079274
-
New roles of NCX in glial cells: activation of microglia in ischemia and differentiation of oligodendrocytes.Adv Exp Med Biol. 2013;961:307-16. doi: 10.1007/978-1-4614-4756-6_26. Adv Exp Med Biol. 2013. PMID: 23224890 Review.
Cited by
-
Structural insight into the allosteric inhibition of human sodium-calcium exchanger NCX1 by XIP and SEA0400.EMBO J. 2024 Jan;43(1):14-31. doi: 10.1038/s44318-023-00013-0. Epub 2023 Dec 15. EMBO J. 2024. PMID: 38177313 Free PMC article.
-
The hypoxia sensitive metal transcription factor MTF-1 activates NCX1 brain promoter and participates in remote postconditioning neuroprotection in stroke.Cell Death Dis. 2021 Apr 30;12(5):423. doi: 10.1038/s41419-021-03705-9. Cell Death Dis. 2021. PMID: 33931586 Free PMC article.
-
The dual face of glutamate: from a neurotoxin to a potential survival factor-metabolic implications in health and disease.Cell Mol Life Sci. 2019 Apr;76(8):1473-1488. doi: 10.1007/s00018-018-3002-x. Epub 2019 Jan 1. Cell Mol Life Sci. 2019. PMID: 30599069 Free PMC article. Review.
-
Emerging perspectives of copper-mediated transcriptional regulation in mammalian cell development.Metallomics. 2024 Oct 4;16(10):mfae046. doi: 10.1093/mtomcs/mfae046. Metallomics. 2024. PMID: 39375833 Free PMC article. Review.
-
HDAC4 and HDAC5 form a complex with DREAM that epigenetically down-regulates NCX3 gene and its pharmacological inhibition reduces neuronal stroke damage.J Cereb Blood Flow Metab. 2020 Oct;40(10):2081-2097. doi: 10.1177/0271678X19884742. Epub 2019 Nov 7. J Cereb Blood Flow Metab. 2020. PMID: 31696766 Free PMC article.
References
-
- Nicoll DA, Longoni S, Philipson KD. Molecular cloning and functional expression of the cardiac sarcolemmal Na(+)-Ca2+ exchanger. Science 1990; 250(4980): 562–565. - PubMed
-
- Li Z, Matsuoka S, Hryshko LV, et al. Cloning of the NCX2 isoform of the plasma membrane Na(+)-Ca2+ exchanger. J Biol Chem 1994; 269(26): 17434–17439. - PubMed
-
- Nicoll DA, Quednau BD, Qui Z, et al. Cloning of a third mammalian Na+-Ca2+ exchanger, NCX3. J Biol Chem 1996; 271(40): 24914–24921. - PubMed
-
- Jeon D, Yang YM, Jeong MJ, et al. Enhanced learning and memory in mice lacking Na+/Ca2+ exchanger 2. Neuron 2003; 38(6): 965–976. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

