Improvement in glycated haemoglobin evaluated by baseline body mass index: a meta-analysis of the liraglutide phase III clinical trial programme
- PMID: 26662611
- PMCID: PMC5067695
- DOI: 10.1111/dom.12617
Improvement in glycated haemoglobin evaluated by baseline body mass index: a meta-analysis of the liraglutide phase III clinical trial programme
Abstract
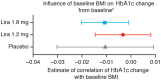
In the liraglutide clinical trial programme, liraglutide 1.2 and 1.8 mg were found to effectively lower glycated haemoglobin (HbA1c) in patients with type 2 diabetes (T2D). It is unknown whether baseline body mass index (BMI) is a predictor of change in HbA1c observed during a clinical trial with liraglutide or placebo treatment. The present meta-analysis of patient-level data, using pooled data from seven phase III trials [LEAD-1-6 and the liraglutide versus sitagliptin trial (LIRA-DPP-4)] for liraglutide 1.2, 1.8 mg and placebo (n = 3222), identified no significant correlation between baseline BMI (<20 kg/m(2) up to 45 kg/m(2) ) and HbA1c reduction for placebo or liraglutide 1.2 mg, and a modest, clinically non-relevant, association for liraglutide 1.8 mg [-0.010 (95% confidence interval -0.020, -0.001)], whereby a 10 kg/m(2) increase in baseline BMI corresponded to 0.10%-point (1.1 mmol/mol) greater HbA1c reduction. In summary, reductions in HbA1c obtained during clinical trials with liraglutide or placebo treatment were independent of baseline BMI.
Keywords: body mass index; glycaemic control; liraglutide; meta-analysis; type 2 diabetes.
© 2015 The Authors. Diabetes, Obesity and Metabolism published by John Wiley & Sons Ltd.
Figures

References
-
- Marre M, Shaw J, Brandle M et al. Liraglutide, a once‐daily human GLP‐1 analogue, added to a sulphonylurea over 26 weeks produces greater improvements in glycaemic and weight control compared with adding rosiglitazone or placebo in subjects with Type 2 diabetes (LEAD‐1 SU). Diabet Med 2009; 26: 268–278. - PMC - PubMed
-
- Garber A, Henry R, Ratner R et al. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD‐3 Mono): a randomised, 52‐week, phase III, double‐blind, parallel‐treatment trial. Lancet 2009; 373: 473–481. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

