Ring expansions of acyloxy nitroso compounds
- PMID: 26663984
- PMCID: PMC4671303
- DOI: 10.1016/j.tetlet.2015.09.002
Ring expansions of acyloxy nitroso compounds
Abstract
Treatment of cyclopentanone and cyclobutanone-derived oximes with lead (IV) tetraacetate gives the bright blue acyloxy nitroso compounds, which upon basic hydrolysis yields the ring expansion product cyclic hydroxamic acids in 12-81% yield. Reactions of substituted cyclopentanones provide ring expanded products where the -NOH group regioselectively inserts to the more substituted position and gives a better yield compared to the treatment of the same ketone with a basic solution of Piloty's acid. Reaction of phosphines with acyloxy nitroso compounds generally generates a ring-expanded Beckmann rearrangement product that can be hydrolyzed to the corresponding lactam. Acyloxy nitroso compounds that undergo rapid hydrolysis to HNO do not show this ring expansion reactivity. These results further demonstrate the versatility of acyloxy nitroso compound to yield structurally complex materials.
Keywords: Acyloxy nitroso compounds; Cyclic hydroxamic acids; HNO donors; Organic phosphines; Ring expansion.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




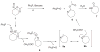

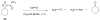
Similar articles
-
Hydrolysis of acyloxy nitroso compounds yields nitroxyl (HNO).J Am Chem Soc. 2006 Aug 2;128(30):9687-92. doi: 10.1021/ja062365a. J Am Chem Soc. 2006. PMID: 16866522
-
Synthesis of cyclic hydroxamic acids through -NOH insertion of ketones.Org Lett. 2009 Oct 15;11(20):4580-3. doi: 10.1021/ol9018198. Org Lett. 2009. PMID: 19743817
-
The chemistry of nitroxyl-releasing compounds.Antioxid Redox Signal. 2011 May 1;14(9):1637-48. doi: 10.1089/ars.2010.3838. Epub 2011 Mar 2. Antioxid Redox Signal. 2011. PMID: 21235345 Free PMC article. Review.
-
New acyloxy nitroso compounds with improved water solubility and nitroxyl (HNO) release kinetics and inhibitors of platelet aggregation.Bioorg Med Chem. 2015 Sep 1;23(17):6069-77. doi: 10.1016/j.bmc.2015.04.023. Epub 2015 Apr 16. Bioorg Med Chem. 2015. PMID: 26228501 Free PMC article.
-
Hydroxamic acids (therapeutics and mechanism): chemistry, acyl nitroso, nitroxyl, reactive oxygen species, and cell signaling.J Recept Signal Transduct Res. 2011 Feb;31(1):10-9. doi: 10.3109/10799893.2010.497152. Epub 2010 Jun 30. J Recept Signal Transduct Res. 2011. PMID: 20590405 Review.
Cited by
-
Total Synthesis of (-)-Flueggeacosine C.Org Lett. 2024 Sep 6;26(35):7341-7346. doi: 10.1021/acs.orglett.4c02516. Epub 2024 Aug 23. Org Lett. 2024. PMID: 39177183 Free PMC article.
-
Total Synthesis of (±)-Phyllantidine: Development and Mechanistic Evaluation of a Ring Expansion for Installation of Embedded Nitrogen-Oxygen Bonds.Angew Chem Int Ed Engl. 2020 Jun 8;59(24):9757-9766. doi: 10.1002/anie.202003829. Epub 2020 May 8. Angew Chem Int Ed Engl. 2020. PMID: 32271982 Free PMC article.
-
Synthetic Potential of Regio- and Stereoselective Ring Expansion Reactions of Six-Membered Carbo- and Heterocyclic Ring Systems: A Review.Int J Mol Sci. 2023 Apr 3;24(7):6692. doi: 10.3390/ijms24076692. Int J Mol Sci. 2023. PMID: 37047665 Free PMC article. Review.
-
New Chemo-, Regio- and Stereoselective Reactions and Methods in Organic Synthesis.Int J Mol Sci. 2024 Dec 14;25(24):13409. doi: 10.3390/ijms252413409. Int J Mol Sci. 2024. PMID: 39769175 Free PMC article.
-
Recent advances in the chemical biology of nitroxyl (HNO) detection and generation.Nitric Oxide. 2016 Jul 1;57:1-14. doi: 10.1016/j.niox.2016.04.006. Epub 2016 Apr 20. Nitric Oxide. 2016. PMID: 27108951 Free PMC article. Review.
References
-
- Samuni U, Samuni Y, Goldstein S. J Am Chem Soc. 2010;132:8428–8432. - PubMed
-
- Irvine JC, Ritchie RH, Favaloro JL, Andrews KL, Widdop RE, Kemp-Harper BK. Trends Pharmacol Sci. 2008;29:601–608. - PubMed
-
- Fukuto JM, Bartberger MD, Dutton AS, Paolocci N, Wink DA, Houk KN. Chem Res Toxicol. 2005;18:790–801. - PubMed
-
- Miranda KM. Coord Chem Rev. 2005;249:433–455.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
