Graphic Warning Labels Elicit Affective and Thoughtful Responses from Smokers: Results of a Randomized Clinical Trial
- PMID: 26672982
- PMCID: PMC4684406
- DOI: 10.1371/journal.pone.0142879
Graphic Warning Labels Elicit Affective and Thoughtful Responses from Smokers: Results of a Randomized Clinical Trial
Abstract
Objective: Observational research suggests that placing graphic images on cigarette warning labels can reduce smoking rates, but field studies lack experimental control. Our primary objective was to determine the psychological processes set in motion by naturalistic exposure to graphic vs. text-only warnings in a randomized clinical trial involving exposure to modified cigarette packs over a 4-week period. Theories of graphic-warning impact were tested by examining affect toward smoking, credibility of warning information, risk perceptions, quit intentions, warning label memory, and smoking risk knowledge.
Methods: Adults who smoked between 5 and 40 cigarettes daily (N = 293; mean age = 33.7), did not have a contra-indicated medical condition, and did not intend to quit were recruited from Philadelphia, PA and Columbus, OH. Smokers were randomly assigned to receive their own brand of cigarettes for four weeks in one of three warning conditions: text only, graphic images plus text, or graphic images with elaborated text.
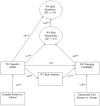
Results: Data from 244 participants who completed the trial were analyzed in structural-equation models. The presence of graphic images (compared to text-only) caused more negative affect toward smoking, a process that indirectly influenced risk perceptions and quit intentions (e.g., image->negative affect->risk perception->quit intention). Negative affect from graphic images also enhanced warning credibility including through increased scrutiny of the warnings, a process that also indirectly affected risk perceptions and quit intentions (e.g., image->negative affect->risk scrutiny->warning credibility->risk perception->quit intention). Unexpectedly, elaborated text reduced warning credibility. Finally, graphic warnings increased warning-information recall and indirectly increased smoking-risk knowledge at the end of the trial and one month later.
Conclusions: In the first naturalistic clinical trial conducted, graphic warning labels are more effective than text-only warnings in encouraging smokers to consider quitting and in educating them about smoking's risks. Negative affective reactions to smoking, thinking about risks, and perceptions of credibility are mediators of their impact.
Trial registration: Clinicaltrials.gov NCT01782053.
Conflict of interest statement
Figures


References
-
- Centers for Disease Control and Prevention (2014). Smoking & tobacco use: Fast facts. Retrieved December 12, 2014, 2014, from http://www.cdc.gov/tobacco/data_statistics/fact_sheets/fast_facts/
-
- Canadian Cancer Society. Cigarette Package Health Warnings: International Status Report, Fourth Edition, September 2014.
-
- R. J. Reynolds Tobacco Co. v. Food and Drug Administration, No. 11–5332 (United States Court of Appeals for the District of Columbia, 2012).
-
- R.J. Reynolds Tobacco Company, et al. v. U.S. Food and Drug Administration, Civil Case No. 11–1482 (RJL) (D.C.D.C., Nov. 7, 2011).
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

