Recombinant factor VIIa concentrate versus plasma-derived concentrates for treating acute bleeding episodes in people with haemophilia and inhibitors
- PMID: 26677005
- PMCID: PMC7137678
- DOI: 10.1002/14651858.CD004449.pub4
Recombinant factor VIIa concentrate versus plasma-derived concentrates for treating acute bleeding episodes in people with haemophilia and inhibitors
Abstract
Background: In people with haemophilia, therapeutic clotting agents might be recognised as a foreign protein and induce anti-factor VIII antibodies, known as 'inhibitors'. Drugs insensitive to such antibodies, either recombinant or plasma-derived, are called factor VIII 'by-passing' agents and used for treatment of bleeding in people with inhibitors.
Objectives: To determine the clinical effectiveness of recombinant factor VIIa concentrate compared to plasma-derived concentrates for treating acute bleeding episodes in people with haemophilia and inhibitors.
Search methods: We searched the Cochrane Cystic Fibrosis and Genetic Disorders Group Coagulopathies Trials Register which comprises references identified from comprehensive electronic database searches and handsearches of relevant journals and abstract books of conference proceedings.Date of the most recent search of the Group's Coagulopathies Trials Register: 23 September 2015.
Selection criteria: Randomised and quasi-randomised controlled clinical trials comparing recombinant factor VIIa concentrate to human plasma-derived concentrates (high-dose human or recombinant factor VIII or factor IX concentrate; non-activated prothrombin complex concentrates; activated prothrombin complex concentrates) in people with haemophilia. Comparisons with animal-derived products were excluded.
Data collection and analysis: Two authors independently assessed the trials (eligibility and risk of bias) and extracted data. No combined meta-analyses were performed due to the unavailability of outcomes and comparisons common to the included trials.
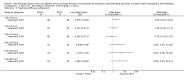
Main results: A total of 15 trials were identified, two of which (with data for a total of 69 participants) were eligible for analysis. Both trials showed methodological flaws and did not show superiority of one treatment over the other. Both the treatments showed that recombinant factor VIIa and activated prothrombin complex concentrate appeared to have a similar haemostatic effect in both trials, without increasing thromboembolic risk.
Authors' conclusions: Based on the separate analysis of the two available randomised trials, recombinant factor VIIa and activated prothrombin complex concentrate were found to be similar in efficacy and safety. However, there is a need for further, well-designed, adequately-powered, randomised controlled trials to assess the relative benefits and risks of using recombinant factor VIIa compared to human plasma-derived concentrates in people with haemophilia with inhibitors. It is advisable that researchers in the field define commonly agreed objective outcome measures in order to enable the pooling of their results, thus increasing the power of comparisons. To date, data could not be combined in a formal meta-analysis. For the same reason reporting concordant and discordant pairs in cross-over trials is recommended.
Conflict of interest statement
Davide Matino declares no conflict of interest.
Michael Makris has received fees for consultancy and sponsorship to attend scientific meetings from Baxter Healthcare and NovoNordisk.
Kerry Dwan declares no conflict of interest.
Roberto D'Amico declares no conflict of interest.
Alfonso Iorio has acted as paid lecturer and has been reimbursed for participation at International Congresses by both NovoNordisk and Baxter Healthcare.
"When originally published, the authors declared the above conflicts of interest. From 07 April 2020, the following conflicts of interest were declared. These conflicts applied during the period that the review was in preparation."Clarification statement added from the Co‐ordinating Editor on 07 April 2020: This review was found by the Cochrane Funding Arbiters, post‐publication, to be noncompliant with the
Figures





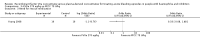



Update of
-
Recombinant Factor VIIa concentrate versus plasma derived concentrates for the treatment of acute bleeding episodes in people with haemophilia and inhibitors.Cochrane Database Syst Rev. 2010 Aug 4;(8):CD004449. doi: 10.1002/14651858.CD004449.pub3. Cochrane Database Syst Rev. 2010. Update in: Cochrane Database Syst Rev. 2015 Dec 16;(12):CD004449. doi: 10.1002/14651858.CD004449.pub4. PMID: 20687076 Updated.
References
References to studies included in this review
Astermark 2007 {published data only}
-
- Astermark J, Donfiled SM, DiMichele DM, Gringeri A, Gilbert SA, Waters J, et al. A randomized comparison of bypassing agents in hemophilia complicated by an inhibitor: the FEIBA NovoSeven Comparative (FENOC) study. Blood 2007;109(2):546‐51. - PubMed
-
- Carllson KS, Astermark J, Donfield SM, Berntorp E. Health‐Economic analysis of alternative bypassing agent in haemophilia complicated by an inhibitor ‐ the FEIBA NovoSeven comparative study (FENOC). Blood 2006;108:457. - PubMed
Young 2008 {published data only}
-
- Young G, Shaffer FE, Rojas P, Seremetis S. Single 270 mcg/kg ‐dose rFVIIa versus standard 90 mcg/kg ‐ dose rFVIIa and APCC for home treatment of joint bleeds in haemophilia patients with inhibitors: a randomized comparison. Haemophilia 2008;14(2):287‐94. - PubMed
References to studies excluded from this review
Chuansumrit 2000 {published data only}
-
- Chuansumrit A, Isarangkura P, Angchaisuksiri P, Sriudomporn N, Tanpowpong K, Hathirat P, et al. Controlling acute bleeding episodes with recombinant factor VIIa in haemophiliacs with inhibitor: continuous infusion and bolus injection. Haemophilia 2000;6(2):61‐5. - PubMed
de Paula 2012 {published data only}
-
- Paula EV, Kavakli K, Mahlangu J, Ayob Y, Lentz SR, Morfini M, et al. Recombinant factor VIIa analog (vatreptacog alfa [activated]) for treatment of joint bleeds in hemophilia patients with inhibitors: a randomized controlled trial. Journal of Thrombosis and Haemostasis 2012;10(1):81‐9. - PubMed
Kavakli 2006 {published data only}
-
- Kavakli K, Makris M, Zulfikar B, Erhardtsen E, Abrams ZS, Kent G. Home treatment of haemarthroses using a single dose regimen of recombinant activated factor seven in patients with haemophilia and inhibitors. A multi‐center, randomized, double‐blind, cross‐over trial. Thrombosis and Haemostasis 2006;95(4):600‐5. - PubMed
Ljung 2013 {published data only}
-
- Ljung R, Karim FA, Saxena K, Suzuki T, Arkhammar P, Rosholm A, et al. 40K glycoPEGylated, recombinant FVIIa: 3‐month, double‐blind, randomized trial of safety, pharmacokinetics and preliminary efficacy in hemophilia patients with inhibitors. Journal of Thrombosis and Haemostasis 2013;11(7):1260‐8. - PubMed
Lusher 1998 {published data only}
-
- Lusher JM, Roberts HR, Davignon G, Joist JH, Smith H, Shapiro A, et al. A randomized, double‐blind comparison of two dosage levels of recombinant factor VIIa in the treatment of joint, muscle and mucocutaneous haemorrhages in persons with haemophilia A and B, with and without inhibitors. rFVIIa Study Group. Haemophilia 1998;4(6):790‐8. - PubMed
Mahlangu 2012 {published data only (unpublished sought but not used)}
-
- Mahlangu JN, Coetzee MJ, Laffan M, Windyga J, Yee TT, Schroeder J, et al. Phase I, randomized, double‐blind, placebo‐controlled, single‐dose escalation study of the recombinant factor VIIa variant BAY 86‐6150 in hemophilia. Journal of Thrombosis and Haemostasis 2012;10(5):773‐80. - PubMed
NCT00108758 {unpublished data only}
-
- NCT00108758. Exploratory, multicenter, randomized, double‐blind, uncontrolled trial evaluating the efficacy of activated rFVII (Novoseven) in secondary bleeding prophylaxis in congenital hemophilia A or B patients with inhibitors. clinicaltrials.gov/show/ NCT00108758 (accessed 23 October 2013).
NCT01561391 {unpublished data only}
-
- NCT01561391. Safety and efficacy of activated human recombinant FVIIa in patients with inhibitors during and after major surgery. clinicaltrials.gov/show/NCT01561391(accessed 5 December 2012) (accessed 5 December 2012).
Pruthi 2007 {published data only}
-
- Pruthi RK, Matthew P. Valentino LA, Sumner MJ, Seremetis S, Hoots WK. Haemostatic efficacy and safety of bolus and continuous infusion of recombinant factor VIIa are comparable in haemophilia patients with inhibitors undergoing major surgery. Results from an open label, randomized multicenter trial. Thrombosis and Haemostasis 2007;98(4):726‐32. - PubMed
Santagostino 2006 {published data only}
-
- Santagostino E, Mancuso ME, Rocino A, Mancuso G, Scaraggi F, Mannucci PM. A prospective randomized trial of high and standard dosages of recombinant factor VIIa for treatment of hemarthroses in hemophiliacs with inhibitors. Journal of Thrombosis and Haemostasis 2006;4(2):367‐71. - PubMed
Seremetis 1994 {published data only}
-
- Seremetis SV. The clinical use of factor VIIa in the treatment of factor VIII inhibitor patients. Seminars in Hematology 1994;31(2 Suppl 4):53‐5. - PubMed
Shapiro 1998 {published data only}
-
- Cooper HA, Gilchrist GS, Hoots WK, Shapiro A. Comparison of two doses of recombinant factor VIIa for producing hemostasis during and after surgery in patients with hemophilia A or B and inhibitors and patients with acquired inhibitors. [abstract]. Blood 1997;90(10 Suppl 1 Pt 1):600a.
-
- Cooper HA, Hoots WK, Shapiro A. Comparison of two doses of recombinant factor Vlla (rFVlla) for producing hemostasis during and after surgery in patients (PTS) with hemophilia A or B and inhibitors and PTS with acquired inhibitors [abstract]. Thrombosis and Haemostasis 1997;Suppl:167‐8.
-
- Shapiro AD, Gilchrist GS, Hoots WK, Cooper HA, Gastineau DA. Prospective, randomised trial of two doses of rFVIIa (NovoSeven) in haemophilia patients with inhibitors undergoing surgery. Thrombosis and Haemostasis 1998;80(5):773‐8. - PubMed
Villar 2004 {published data only}
-
- Villar A, Aronis S, Morfini M, Santagostino E, Auerswald G, Thomsen HF, et al. Pharmacokinetics of activated recombinant coagulation factor VII (NovoSeven) in children vs. adults with haemophilia A. Haemophilia 2004;10(4):352‐9. - PubMed
Additional references
Athale 2014
Becker 1993
-
- Becker MP, Balagtas CC. Marginal modelling of binary cross‐over data. Biometrics 1993;49(4):997‐1009. - PubMed
Dimichele 2002
-
- Dimichele D. Inhibitors: resolving diagnostic and therapeutic dilemmas. Haemophilia 2002;8(3):280‐7. - PubMed
DiMichele 2007
Elbourne 2002
-
- Elbourne DR, Altman DG, Higgins JPT, Curtin F, Worthington HV, Vail A. Meta‐analysis involving cross‐over trials: methodological issues. International Journal of Epidemiology 2002;31(1):140‐9. - PubMed
Ewenstein 1997
-
- Ewenstein BM, Takemoto C, Warrier I, Lusher J, Saidi P, Eisele J, et al. Nephrotic syndrome as a complication of immune tolerance in hemophilia B. Blood 1997;89(3):1115‐6. - PubMed
Goodeve 2003
-
- Goodeve AC, Peake IR. The molecular basis of hemophilia a: genotype‐phenotype relationships and inhibitor development. Seminars in Thrombosis and Hemostasis 2003;29(1):23‐30. - PubMed
Hay 2000
-
- Hay CRM, Baglin TP, Collins PW, Hill FGH, Keeling DM. The diagnosis and management of factor VIII and IX inhibitors: a guideline from the UK Haemophilia Centre Doctors' Organization (UKHCDO). British Journal of Haematology 2000;111(1):78‐90. - PubMed
Hedner 2000
-
- Hedner U. Recombinant coagulation factor VIIa: from the concept to clinical application in hemophilia treatment in 2000. Seminars in Thrombosis and Hemostasis 2000;26(4):363‐6. - PubMed
Higgins 2003
Knight 2009
Kreuz 2002
-
- Kreuz W, Ettingshausen CE, Zyschka A, Oldenberg J, Saguer IM, Ehrenforth S, et al. Inhibitor development in previously untreated patients with hemophilia A: a prospective long‐term follow‐up comparing plasma‐derived and recombinant products. Seminars in Thrombosis and Hemostasis 2002;28(3):285‐90. - PubMed
Lloyd Jones 2003
-
- Lloyd Jones M, Wight J, Paisley S, Knight C. Control of bleeding in patients with haemophilia A with inhibitors: a systematic review. Haemophilia 2003;9(4):464‐520. - PubMed
Mannucci 2001
-
- Mannucci PM, Tuddenham EG. The hemophilias ‐ from royal genes to gene therapy. New England Journal of Medicine 2001;344(23):1773‐9. - PubMed
Rizza 2001
-
- Rizza CR, Spooner RJ, Giangrande PL, UK Haemophilia Centre Doctors' Organization (UKCDO). Treatment of haemophilia in the United Kingdom 1981‐1996. Haemophilia 2001;7(4):349‐59. - PubMed
Thomas 1977
Treur 2009
-
- Treur MJ, McCracken F, Heeg B, Joshi AV, Botteman MF, Charro F, et al. Efficacy of recombinant activated factor seven vs activated prothrombin complex concentrates for patients suffering from haemophilia complicated with inhibitors: a Bayesian meta‐regression. Haemophilia 2009;15(2):420‐36. [DOI: 10.1111/j.1365-2516.2008.01956.x] - DOI - PubMed
UKHCDO 2003
-
- United Kingdom Haemophilia Doctors' Organisation. Guidelines on the selection and use of therapeutic products to treat haemophilia and other hereditary bleeding disorders. Haemophilia 2003;9(1):1‐13. - PubMed
White 2001
-
- White GC 2nd, Rosendaal F, Aledort LM, Lusher JM, Rothschild C, Ingerslev J, et al. Definitions in hemophilia. Recommendation of the scientific subcommittee on factor VIII and factor IX of the scientific and standardization committee of the International Society on Thrombosis and Haemostasis. Thrombosis and Haemostasis 2001;85(3):560. - PubMed
Yee 1999
-
- Yee TT, Pasi KJ, Lilley PA, Lee CA. Factor VIII inhibitors in haemophiliacs: a single‐centre experience over 34 years, 1964‐97. British Journal of Haematology 1999;104(4):909‐14. - PubMed
References to other published versions of this review
Iorio 2010
-
- Iorio A, Matino D, D'Amico R, Makris M. Recombinant Factor VIIa concentrate versus plasma derived concentrates for the treatment of acute bleeding episodes in people with haemophilia and inhibitors. Cochrane Database of Systematic Reviews 2010, Issue 8. [DOI: 10.1002/14651858.CD004449.pub3] - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

