Profile of nintedanib in the treatment of solid tumors: the evidence to date
- PMID: 26677336
- PMCID: PMC4677757
- DOI: 10.2147/OTT.S78805
Profile of nintedanib in the treatment of solid tumors: the evidence to date
Abstract
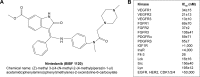
Angiogenesis is an essential process for tumor growth and metastasis, and remains a promising therapeutic target process in cancer treatment for several cancer types. Bevacizumab, a monoclonal antibody that targets vascular endothelial growth factor (VEGF), was the first antiangiogenic agent approved for cancer therapy. Novel antiangiogenic agents, such as sunitinib, sorafenib, pazopanib, or vandetanib that target additional proangiogenic signaling pathways beyond VEGF, have also been approved for the treatment of various malignant diseases. While most of these agents are approved in combination with cytotoxic chemotherapy for indications including metastatic colorectal cancer, non-small-cell lung cancer, breast cancer, renal cell carcinoma (RCC), and gastric cancer, some are used as approved monotherapy for advanced RCC, hepatocellular carcinoma and medullary thyroid cancer. Major challenges to the success of antiangiogenic therapy include associated toxicity risks, limitation of efficacy through the possible development of resistance and induction or promotion of metastatic progression. Nintedanib (formally known as BIBF 1120) is a triple angiokinase inhibitor of VEGF, fibroblast growth factor, platelet-derived growth factor signaling with lesser activity against RET, Flt-3, and Src. Through this unique targeting profile nintedanib has demonstrated significant antitumor activity in several tumor types in preclinical studies. Nintedanib has also shown promising clinical efficacy in combination with docetaxel and has been approved for treating patients with locally advanced and metastatic non-small-cell lung cancer in Europe. Nintedanib has also been found to be clinically promising in terms of efficacy and safety in several other solid tumors including ovarian cancer (Phase III), RCC (Phase II), and prostate cancer (Phase II). This review article provides a comprehensive summary of the preclinical and clinical efficacy of nintedanib in the treatment of solid tumors.
Keywords: BIBF 1120; VEGF; angiogenesis; nintedanib; tyrosine kinase inhibitors.
Figures
Similar articles
-
BIBF 1120/ nintedanib : a new triple angiokinase inhibitor-directed therapy in patients with non-small cell lung cancer.Expert Opin Investig Drugs. 2013 Aug;22(8):1081-8. doi: 10.1517/13543784.2013.812630. Epub 2013 Jun 22. Expert Opin Investig Drugs. 2013. PMID: 23790097 Review.
-
Nintedanib in non-small cell lung cancer: from preclinical to approval.Ther Adv Respir Dis. 2015 Aug;9(4):164-72. doi: 10.1177/1753465815579608. Epub 2015 Apr 7. Ther Adv Respir Dis. 2015. PMID: 25855060 Review.
-
Nintedanib, a novel triple angiokinase inhibitor for the treatment of non-small cell lung cancer.Drugs Today (Barc). 2015 Jun;51(6):357-66. doi: 10.1358/dot.2015.51.6.2319324. Drugs Today (Barc). 2015. PMID: 26261849 Review.
-
Exploring the Benefit/Risk Associated with Antiangiogenic Agents for the Treatment of Non-Small Cell Lung Cancer Patients.Clin Cancer Res. 2017 Mar 1;23(5):1137-1148. doi: 10.1158/1078-0432.CCR-16-1968. Epub 2016 Dec 9. Clin Cancer Res. 2017. PMID: 27940520 Review.
-
Nintedanib: A Review of Its Use as Second-Line Treatment in Adults with Advanced Non-Small Cell Lung Cancer of Adenocarcinoma Histology.Target Oncol. 2015 Jun;10(2):303-10. doi: 10.1007/s11523-015-0367-8. Target Oncol. 2015. PMID: 25894578 Review.
Cited by
-
RNA splicing: Novel star in pulmonary diseases with a treatment perspective.Acta Pharm Sin B. 2025 May;15(5):2301-2322. doi: 10.1016/j.apsb.2025.03.023. Epub 2025 Mar 13. Acta Pharm Sin B. 2025. PMID: 40487667 Free PMC article. Review.
-
FGF/FGFR signaling in health and disease.Signal Transduct Target Ther. 2020 Sep 2;5(1):181. doi: 10.1038/s41392-020-00222-7. Signal Transduct Target Ther. 2020. PMID: 32879300 Free PMC article. Review.
-
Targeting neurokinin-3 receptor: a novel anti-angiogenesis strategy for cancer treatment.Oncotarget. 2017 Jun 20;8(25):40713-40723. doi: 10.18632/oncotarget.17250. Oncotarget. 2017. PMID: 28489574 Free PMC article.
-
The State-of-the-Art of Phase II/III Clinical Trials for Targeted Pancreatic Cancer Therapies.J Clin Med. 2021 Feb 3;10(4):566. doi: 10.3390/jcm10040566. J Clin Med. 2021. PMID: 33546207 Free PMC article. Review.
-
Src Tyrosine Kinase Inhibitors: New Perspectives on Their Immune, Antiviral, and Senotherapeutic Potential.Front Pharmacol. 2019 Sep 18;10:1011. doi: 10.3389/fphar.2019.01011. eCollection 2019. Front Pharmacol. 2019. PMID: 31619990 Free PMC article. Review.
References
-
- Folkman J. Tumor angiogenesis: therapeutic implications. N Engl J Med. 1971;285(21):1182–1186. - PubMed
-
- Moreira IS, Fernandes PA, Ramos MJ. Vascular endothelial growth factor (VEGF) inhibition – a critical review. Anticancer Agents Med Chem. 2007;7(2):223–245. - PubMed
-
- Ferrara N. Vascular endothelial growth factor as a target for anticancer therapy. Oncologist. 2004;9(Suppl 1):2–10. - PubMed
-
- Shojaei F, Ferrara N. Antiangiogenic therapy for cancer: an update. Cancer J. 2007;13(6):345–348. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

