Low-molecular-weight heparins for managing vaso-occlusive crises in people with sickle cell disease
- PMID: 26684281
- PMCID: PMC7087472
- DOI: 10.1002/14651858.CD010155.pub3
Low-molecular-weight heparins for managing vaso-occlusive crises in people with sickle cell disease
Abstract
Background: Sickle cell disease is one of the most common and severe genetic disorders in the world. It can be broadly divided into two distinct clinical phenotypes characterized by either haemolysis or vaso-occlusion. Pain is the most prominent symptom of vaso-occlusion, and hypercoagulability is a well-established pathogenic phenomenon in people with sickle cell disease. Low-molecular-weight heparins might control this hypercoagulable state through their anticoagulant effect. This is an update of a previously published version of this review.
Objectives: To assess the effects of low-molecular-weight heparins for managing vaso-occlusive crises in people with sickle cell disease.
Search methods: We searched the Cochrane Cystic Fibrosis and Genetic Disorders Group Haemoglobinopathies Trials Register comprising references identified from comprehensive electronic database searches. We also searched abstract books of conference proceedings and several online trials registries for ongoing trials.Date of the last search of the Cochrane Cystic Fibrosis and Genetic Disorders Group Haemoglobinopathies Trials Register: 28 September 2015.
Selection criteria: Randomised controlled clinical trials and controlled clinical trials that assessed the effects of low-molecular-weight heparins in the management of vaso-occlusive crises in people with sickle cell disease.
Data collection and analysis: Study selection, data extraction, assessment of risk of bias and analyses were carried out independently by the two review authors.
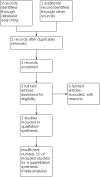
Main results: Two studies comprising 287 participants were included. One study (with an overall unclear to high risk of bias) involved 253 participants and the quality of the evidence for most outcomes was very low. This study, reported that pain severity at day two and day three was lower in the tinzaparin group than in the placebo group (P < 0.01, analysis of variance (ANOVA)) and additionally at day 4 (P < 0.05 (ANOVA)). Thus tinzaparin resulted in more rapid resolution of pain, as measured with a numerical pain scale. The mean difference in duration of painful crises was statistically significant at -1.78 days in favour of the tinzaparin group (95% confidence interval -1.94 to -1.62). Participants treated with tinzaparin had statistically significantly fewer hospitalisation days than participants in the group treated with placebo, with a mean difference of -4.98 days (95% confidence interval -5.48 to -4.48). Two minor bleeding events were reported as adverse events in the tinzaparin group, and none were reported in the placebo group. The second study (unclear risk of bias) including 34 participants and was a conference abstract with limited data and only addressed one of the predefined outcomes of the review; i.e. pain intensity. After one day pain intensity reduced more, as reported on a visual analogue scale, in the dalteparin group than in the placebo group, mean difference -1.30 (95% confidence interval -1.60 to -1.00), with the quality of evidence rated very low. The most important reasons for downgrading the quality of evidence were serious risk of bias and imprecision (due to low sample size or low occurrence of events).
Authors' conclusions: Based on the results of two studies, evidence is incomplete to support or refute the effectiveness of low-molecular-weight heparins in people with sickle cell disease. Vaso-occlusive crises are extremely debilitating for sufferers of sickle cell disease; therefore well-designed placebo-controlled studies with other types of low-molecular-weight heparins, and in participants with different genotypes of sickle cell disease, still need to be carried out to confirm or dismiss the results of this single study.
Conflict of interest statement
No financial conflicts of interest have been reported, and the authors declare that they do not have any associations with any parties who may have vested interests in the results of this review.
Figures






Update of
-
Low-molecular-weight heparins for managing vaso-occlusive crises in people with sickle cell disease.Cochrane Database Syst Rev. 2013 Jun 12;(6):CD010155. doi: 10.1002/14651858.CD010155.pub2. Cochrane Database Syst Rev. 2013. Update in: Cochrane Database Syst Rev. 2015 Dec 18;(12):CD010155. doi: 10.1002/14651858.CD010155.pub3. PMID: 23760785 Updated.
References
References to studies included in this review
Qari 2007 {published data only}
-
- Qari MH, Aljaouni SK, Alardawi MS, Fatani H, Alsayes FM, Zografos P, et al. Reduction of painful vaso-occlusive crisis of sickle cell anaemia by tinzaparin in a double-blind randomized trial. Thrombosis and Haemostasis 2007;98(2):392-6. [PMID: PMID: 17721622] - PubMed
-
- Qari MH, Mousa S, Alsaigh MA, Zografos P, Aljaouni SK, Fatani H, et al. Tinzaparin in the management of painful vaso-occlusive crisis of sickle cell anaemia. Blood 2005;106(11):Abstract no: 2340.
Shah 2013 {unpublished data only}
-
- NCT01419977. Treatment of sickle cell patients hospitalized in pain crisis with prophylactic dose low-molecular-weight heparin (LMWH) versus placebo. www.clinicaltrials.gov/show/NCT01419977 (accessed 01 March 2013).
-
- Shah N, Willen S, Telen MJ, Ortel TL. Prophylactic dose low molecular weight heparin (dalteparin) for treatment of vaso-occlusive pain crisis in patients with sickle cell disease [abstract]. Blood 2013;122(21):2241. [CENTRAL: 983639]
Additional references
Al Hajeri 2008
Ataga 2012
Austin 2007
-
- Austin H, Key NS, Benson JM, Lally C, Dowling NF, Whitsett C, et al. Sickle cell trait and the risk of venous thromboembolism among blacks. Blood 2007;110(3):908-12. [PMID: PMID: 17409269] - PubMed
Austin 2009
-
- Austin H, Lally C, Benson JM, Whitsett C, Hooper WC, Key NS. Hormonal contraception, sickle cell trait, and risk for venous thromboembolism among African American women. American Journal of Obstetrics and Gynecology 2009;200(6):620.e1-3. [PMID: PMID: 19306959] - PubMed
Ballas 2010
Ballas 2012
Ballas 2013
-
- Ballas SK. Sickle cell anemia. http://pier.acponline.org/physicians/diseases/d905/d905.html (accessed 28 February 2013):1-93.
Brown 2006
Bunn 2010
-
- Bunn HF, Nathan DG, Dover GJ, Hebbel RP, Platt OS, Rosse WF, et al. Pulmonary hypertension and nitric oxide depletion in sickle cell disease. Blood 2010;116(5):687-92. [PMID: ] - PubMed
Carr 2007
-
- Carr JA, Cho JS. Low molecular weight heparin suppresses tumor necrosis factor expression from deep vein thrombosis. Annals of Vascular Surgery 2007;21(1):50-5. [PMID: PMID: 17349336] - PubMed
Davies 2012
De Franceschi 2009
De Franceschi 2011
-
- De Franceschi L, Cappellini MD, Olivieri O. Thrombosis and sickle cell disease. Seminars in Thrombosis and Hemostasis 2011;37(3):226-36. [PMID: PMID: 21455857] - PubMed
de Montalembert 2008
-
- Montalembert M. Management of sickle cell disease. BMJ 2008;337:a1397. [PMID: PMID: 18779222] - PubMed
Gladwin 2012
Goldsmith 2012
Higgins 2003
Higgins 2011a
-
- Higgins JPT, Altman DG. Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Green S (editors). Cochrane Handbook of Systematic Reviews of Interventions. Version 5.1 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane-handbook.org.
Higgins 2011b
-
- Sterne JAC, Egger M, Moher D, on behalf of the Cochrane Bias Methods Group. Chapter 10: Addressing reporting biases. In: Higgins JPT, Green S (editors). Cochrane Handbook of Systematic Reviews of Interventions. Version 5.1 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane-handbook.org.
Higgins 2011c
-
- Deeks JJ, Higgins JPT, Altman DG. Chapter 9: Analysing data and undertaking meta-analyses. In: Higgins JPT, Green S (editors). Cochrane Handbook of Systematic Reviews of Interventions. Version 5.1 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane-handbook.org .
Higgins 2011d
-
- Higgins JPT, Deeks JJ, Altman DG on behalf of the CSMG . Chapter 16: Special topics in statistics. In: Higgins JPT, Green S (editors). Cochrane Handbook of Systematic Reviews of Interventions. Version 5.1 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane-handbook.org.
Hirani 2011
-
- Hirani A, Weibel S, Kane GC. Acute chest syndrome and other pulmonary manifestations of sickle cell disease. Journal of Clinical Outcome Management 2011;18(5):211-21.
Hirsh 1992
-
- Hirsh J, Levine MN. Low molecular weight heparin. Blood 1992;79(1):1-17. [PMID: PMID: 1309422] - PubMed
Hirsh 2001
-
- Hirsh J, Warkentin TE, Shaughnessy SG, Anand SS, Halperin JL, Raschke R, et al. Heparin and low-molecular-weight heparin: mechanisms of action, pharmacokinetics, dosing, monitoring, efficacy, and safety. Chest 2001;119 Suppl 1:64S-94S. [PMID: PMID: 11157643] - PubMed
Hoy 2010
-
- Hoy SM, Scott LJ, Plosker GL. Tinzaparin sodium: a review of its use in the prevention and treatment of deep vein thrombosis and pulmonary embolism, and in the prevention of clotting in the extracorporeal circuit during haemodialysis. Drugs 2010;70(10):1319-47. [PMID: PMID: 20568836] - PubMed
Inati 2009
-
- Inati A. Recent advances in improving the management of sickle cell disease. Blood Reviews 2009;23 Suppl 1:S9-13. [PMID: PMID: 20116638] - PubMed
Inati 2009b
-
- Inati A, Chabtini L, Mounayar M, Taher A. Current understanding in the management of sickle cell disease. Hemoglobin 2009;33 Suppl 1:S107-15. [PMID: PMID: 20001613] - PubMed
Jaywant 2003
-
- Jaywant S, Pai A. A comparative study of pain measurement scales in acute burn patients. Indian Journal of Occupational Therapy 2003;35(3):13-7.
Key 2010
Mousa 2003
-
- Mousa SA, Bozarth J, Barrett JS. Pharmacodynamic properties of the low molecular weight heparin, tinzaparin: effect of molecular weight distribution on plasma tissue factor pathway inhibitor in healthy human subjects. Journal of Clinical Pharmacology 2003;43(7):727-34. [PMID: PMID: 12856386] - PubMed
Mousa 2010
-
- Mousa SA, Al Momen A, Al Sayegh F, Al Jaouni S, Nasrullah Z, Al Saeed H, et al. Management of painful vaso-occlusive crisis of sickle-cell anemia: consensus opinion. Clinical and Applied Thrombosis/Haemostasis 2010;16(4):365-76. [PMID: PMID: 20530056] - PubMed
Rees 2010
-
- Rees DC, Williams TN, Gladwin MT. Sickle-cell disease. Lancet 2010;376(9757):2018-31. [PMID: PMID: 21131035] - PubMed
Review Manager (Revman) 2014 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Schulz 1995
-
- Schulz KF, Chalmers I, Hayes RJ, Altman DG. Empirical evidence of bias. Dimensions of methodological quality associated with estimates of treatment effects in controlled trials. JAMA 1995;273(5):408-12. - PubMed
Schünemann 2013
-
- Schünemann H, Brożek J, Guyatt G, Oxman A, editors. GRADE handbook for grading quality of evidence and strength of recommendations. Updated October 2013. Available from www.guidelinedevelopment.org/handbook, The GRADE Working Group, 2013.
Steinberg 1999
-
- Steinberg MH. Management of sickle cell disease. New England Journal of Medicine 1999;340(13):1021-30. [PMID: PMID: 10099145] - PubMed
Steinberg 2011
-
- Steinberg MH. In the Clinic. Sickle cell disease. Annals of Internal Medicine 2011;155(5):ITC31-15. [PMID: ] - PubMed
WHO 2006
-
- World Health Organization. Sickle-cell anaemia. Report A59/9. Provisional agenda item 11.4. 59th World Health Assembly. https://apps.who.int/gb/ebwha/pdf_files/WHA59/A59_9-en.pdf (accessed 13 March 2012).
References to other published versions of this review
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

