Cryptococcus: from environmental saprophyte to global pathogen
- PMID: 26685750
- PMCID: PMC5019959
- DOI: 10.1038/nrmicro.2015.6
Cryptococcus: from environmental saprophyte to global pathogen
Abstract
Cryptococcosis is a globally distributed invasive fungal infection that is caused by species within the genus Cryptococcus which presents substantial therapeutic challenges. Although natural human-to-human transmission has never been observed, recent work has identified multiple virulence mechanisms that enable cryptococci to infect, disseminate within and ultimately kill their human host. In this Review, we describe these recent discoveries that illustrate the intricacy of host-pathogen interactions and reveal new details about the host immune responses that either help to protect against disease or increase host susceptibility. In addition, we discuss how this improved understanding of both the host and the pathogen informs potential new avenues for therapeutic development.
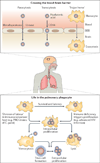
Figures


References
-
- Kwon-Chung JK, Boekhout T, Fell JW, Diaz M. Proposal to conserve the name Cryptococcus gattii against C. hondurianus and C. basillisporus (Basidiomycota, Hymenomycetes, Tremellomycetidae) Taxon. 2002;51:804–806.
-
- Hagen F, et al. Recognition of seven species in the Cryptococcus gattii/Cryptococcus neoformans species complex. Fungal Genet. Biol. 2015;78:16–48. - PubMed
-
- Chowdhary A, Rhandhawa HS, Prakash A, Meis JF. Environmental prevalence of Cryptococcus neoformans and Cryptococcus gattii in India: an update. Crit. Rev. Microbiol. 2012;38:1–16. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

