Prophylactic antibiotics or G(M)-CSF for the prevention of infections and improvement of survival in cancer patients receiving myelotoxic chemotherapy
- PMID: 26687844
- PMCID: PMC7389519
- DOI: 10.1002/14651858.CD007107.pub3
Prophylactic antibiotics or G(M)-CSF for the prevention of infections and improvement of survival in cancer patients receiving myelotoxic chemotherapy
Abstract
Background: Febrile neutropenia (FN) and other infectious complications are some of the most serious treatment-related toxicities of chemotherapy for cancer, with a mortality rate of 2% to 21%. The two main types of prophylactic regimens are granulocyte (macrophage) colony-stimulating factors (G(M)-CSF) and antibiotics, frequently quinolones or cotrimoxazole. Current guidelines recommend the use of colony-stimulating factors when the risk of febrile neutropenia is above 20%, but they do not mention the use of antibiotics. However, both regimens have been shown to reduce the incidence of infections. Since no systematic review has compared the two regimens, a systematic review was undertaken.
Objectives: To compare the efficacy and safety of G(M)-CSF compared to antibiotics in cancer patients receiving myelotoxic chemotherapy.
Search methods: We searched The Cochrane Library, MEDLINE, EMBASE, databases of ongoing trials, and conference proceedings of the American Society of Clinical Oncology and the American Society of Hematology (1980 to December 2015). We planned to include both full-text and abstract publications. Two review authors independently screened search results.
Selection criteria: We included randomised controlled trials (RCTs) comparing prophylaxis with G(M)-CSF versus antibiotics for the prevention of infection in cancer patients of all ages receiving chemotherapy. All study arms had to receive identical chemotherapy regimes and other supportive care. We included full-text, abstracts, and unpublished data if sufficient information on study design, participant characteristics, interventions and outcomes was available. We excluded cross-over trials, quasi-randomised trials and post-hoc retrospective trials.
Data collection and analysis: Two review authors independently screened the results of the search strategies, extracted data, assessed risk of bias, and analysed data according to standard Cochrane methods. We did final interpretation together with an experienced clinician.
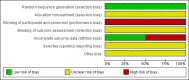
Main results: In this updated review, we included no new randomised controlled trials. We included two trials in the review, one with 40 breast cancer patients receiving high-dose chemotherapy and G-CSF compared to antibiotics, a second one evaluating 155 patients with small-cell lung cancer receiving GM-CSF or antibiotics.We judge the overall risk of bias as high in the G-CSF trial, as neither patients nor physicians were blinded and not all included patients were analysed as randomised (7 out of 40 patients). We considered the overall risk of bias in the GM-CSF to be moderate, because of the risk of performance bias (neither patients nor personnel were blinded), but low risk of selection and attrition bias.For the trial comparing G-CSF to antibiotics, all cause mortality was not reported. There was no evidence of a difference for infection-related mortality, with zero events in each arm. Microbiologically or clinically documented infections, severe infections, quality of life, and adverse events were not reported. There was no evidence of a difference in frequency of febrile neutropenia (risk ratio (RR) 1.22; 95% confidence interval (CI) 0.53 to 2.84). The quality of the evidence for the two reported outcomes, infection-related mortality and frequency of febrile neutropenia, was very low, due to the low number of patients evaluated (high imprecision) and the high risk of bias.There was no evidence of a difference in terms of median survival time in the trial comparing GM-CSF and antibiotics. Two-year survival times were 6% (0 to 12%) in both arms (high imprecision, low quality of evidence). There were four toxic deaths in the GM-CSF arm and three in the antibiotics arm (3.8%), without evidence of a difference (RR 1.32; 95% CI 0.30 to 5.69; P = 0.71; low quality of evidence). There were 28% grade III or IV infections in the GM-CSF arm and 18% in the antibiotics arm, without any evidence of a difference (RR 1.55; 95% CI 0.86 to 2.80; P = 0.15, low quality of evidence). There were 5 episodes out of 360 cycles of grade IV infections in the GM-CSF arm and 3 episodes out of 334 cycles in the cotrimoxazole arm (0.8%), with no evidence of a difference (RR 1.55; 95% CI 0.37 to 6.42; P = 0.55; low quality of evidence). There was no significant difference between the two arms for non-haematological toxicities like diarrhoea, stomatitis, infections, neurologic, respiratory, or cardiac adverse events. Grade III and IV thrombopenia occurred significantly more frequently in the GM-CSF arm (60.8%) compared to the antibiotics arm (28.9%); (RR 2.10; 95% CI 1.41 to 3.12; P = 0.0002; low quality of evidence). Neither infection-related mortality, incidence of febrile neutropenia, nor quality of life were reported in this trial.
Authors' conclusions: As we only found two small trials with 195 patients altogether, no conclusion for clinical practice is possible. More trials are necessary to assess the benefits and harms of G(M)-CSF compared to antibiotics for infection prevention in cancer patients receiving chemotherapy.
Conflict of interest statement
Nicole Skoetz: none known
Julia Bohlius: none known
Ina Monsef: none known
Oliver Blank: none known
Andreas Engert: none known
Jörg Janne Vehreschild: none known
Figures
Update of
-
Prophylactic antibiotics or G-CSF for the prevention of infections and improvement of survival in cancer patients undergoing chemotherapy.Cochrane Database Syst Rev. 2009 Jan 21;(1):CD007107. doi: 10.1002/14651858.CD007107.pub2. Cochrane Database Syst Rev. 2009. Update in: Cochrane Database Syst Rev. 2015 Dec 21;(12):CD007107. doi: 10.1002/14651858.CD007107.pub3. PMID: 19160320 Updated.
References
References to studies included in this review
Schroder 1999 {published data only}
-
- Schroder CP, Vries EG, Mulder NH, Willemse PH, Sleijfer DT, Hospers GA, et al. Prevention of febrile leucopenia after chemotherapy in high‐risk breast cancer patients: no significant difference between granulocyte‐colony stimulating growth factor or ciprofloxacin plus amphotericin B. Journal of Antimicrobial Chemotherapy 1999;43(5):741‐3. - PubMed
Sculier 2001 {published data only}
-
- Sculier JP, Paesmans M, Lecomte J, Cutsem O, Lafitte JJ, Berghmans T, et al. A three‐arm phase III randomised trial assessing, in patients with extensive‐disease small‐cell lung cancer, accelerated chemotherapy with support of haematological growth factor or oral antibiotics. British Journal of Cancer 2001;85(10):1444‐51. - PMC - PubMed
References to studies excluded from this review
Aarts 2013 {published data only}
-
- Aarts MJ, Peters FP, Mandigers CM, Dercksen MW, Stouthard JM, Nortier HJ, et al. Primary granulocyte colony‐stimulating factor prophylaxis during the first two cycles only or throughout all chemotherapy cycles in patients with breast cancer at risk for febrile neutropenia. Journal of Clinical Oncology 2013;31(34):4290‐6. [DOI: 10.1200/JCO.2012.44.6229.] - DOI - PubMed
Alonzo 2002 {published data only}
-
- Alonzo TA, Kobrinsky NL, Aledo A, Lange BJ, Buxton AB, Woods WG. Impact of granulocyte colony‐stimulating factor use during induction for acute myelogenous leukemia in children: a report from the Children's Cancer Group. Journal of Pediatric Hematology/Oncology 2002;24(8):627‐35. - PubMed
Altman 1996 {published data only}
-
- Altman A, Steinherz P, Trigg M, Halpern S, Jhanwar S, Pieters R, et al. A prospective randomized trial of granulocyte colony‐stimulating factor (G‐CSF) during induction or consolidation phases of intensive chemotherapy for acute lymphocytic leukemia (ALL) [abstract]. Journal of Clinical Oncology. 1996:542, Abstract 1760.
Ardizzoni 1994 {published data only}
-
- Ardizzoni A, Venturini M, Sertoli MR, Giannessi PG, Brema F, Danova M, et al. Granulocyte‐macrophage colony‐stimulating factor (GM‐CSF) allows acceleration and dose intensity increase of CEF chemotherapy: a randomised study in patients with advanced breast cancer. British Journal of Cancer 1994;69(2):385‐91. - PMC - PubMed
Attal 1991 {published data only}
-
- Attal M, Schlaifer D, Rubie H, Huguet F, Charlet JP, Bloom E, et al. Prevention of gram‐positive infections after bone marrow transplantation by systemic vancomycin: a prospective, randomized trial. Journal of Clinical Oncology 1991;9(5):865‐70. - PubMed
Bennett 2001 {published data only}
-
- Bennett CL, Hynes D, Godwin J, Stinson TJ, Golub RM, Appelbaum FR, et al. Economic analysis of granulocyte colony stimulating factor as adjunct therapy for older patients with acute myelogenous leukemia (AML): estimates from a Southwest Oncology Group clinical trial. Cancer Investigation 2001;19(6):603‐10. - PubMed
Bishop 2000 {published data only}
-
- Bishop MR, Tarantolo SR, Geller RB, Lynch JC, Bierman PJ, Pavletic ZS, et al. A randomized, double‐blind trial of filgrastim (granulocyte colony‐stimulating factor) versus placebo following allogeneic blood stem cell transplantation. Blood 2000;96(1):80‐5. - PubMed
Björkholm 1999 {published data only}
-
- Björkholm M, Osby E, Hagberg H, Kvaloy S, Teerenhovi L, Myhre J. Randomized trial of r‐metHu granulocyte colony‐stimulating factor (G‐CSF) as adjunct to CHOP or CNOP treatment of elderly patients with aggressive non‐Hodgkin's lymphoma [abstract]. Blood. 1999:599a.
Bradstock 2001 {published data only}
-
- Bradstock K, Matthews J, Young G, Lowenthal R, Baxter H, Cetal A. Effects of glycosylated recombinant human granulocyte colony‐stimulating factor after high‐dose cytarabine‐based induction chemotherapy for adult acute myeloid leukaemia. Leukemia 2001;15(9):1331‐8. - PubMed
Brugger 2009 {published data only}
-
- Brugger W, Bacon P, Lawrinson S, Romieu G. Neutrophil recovery in elderly breast cancer patients receiving adjuvant anthracycline‐containing chemotherapy with pegfilgrastim support. Critical Reviews in Oncology/Hematology 2009;72(3):265‐9. - PubMed
Burton 2006 {published data only}
Carlson 1997 {published data only}
-
- Carlson JW, Fowler JM, Mitchell SK, Carson LF, Mayer AR, Copeland LJ. Chemoprophylaxis with ciprofloxacin in ovarian cancer patients receiving paclitaxel: a randomized trial. Gynecologic Oncology 1997;65(2):325‐9. - PubMed
Chevallier 1995 {published data only}
-
- Chevallier B, Chollet P, Merrouche Y, Roche H, Fumoleau P, Kerbrat P, et al. Lenograstim prevents morbidity from intensive induction chemotherapy in the treatment of inflammatory breast cancer. Journal of Clinical Oncology 1995;13(7):1564‐71. - PubMed
Clarke 1999 {published data only}
-
- Clarke V, Dunstan FD, Webb DK. Granulocyte colony‐stimulating factor ameliorates toxicity of intensification chemotherapy for acute lymphoblastic leukemia. Medical & Pediatric Oncology 1999;32(5):331‐5. - PubMed
Crawford 1997 {published data only}
-
- Crawford J, Kreisman H, Garewal H, Jones SE, Shoemaker D, Pupa MR, et al. The impact of filgrastim schedule variation on hematopoietic recovery post‐chemotherapy. Annals of Oncology 1997;8(11):1117‐24. - PubMed
Cullen 2005 {published data only}
-
- Cullen M, Steven N, Billingham L, Gaunt C, Hastings M, Simmonds P, et al. Antibacterial prophylaxis after chemotherapy for solid tumors and lymphomas. New England Journal of Medicine 2005;353(10):988‐98. - PubMed
Dibenedetto 1995 {published data only}
-
- Dibenedetto SP, Ragusa R, Ippolito AM, Lo NL, Cataldo A, D'Amico S, et al. Assessment of the value of treatment with granulocyte colony‐stimulating factor in children with acute lymphoblastic leukemia: a randomized clinical trial. European Journal of Haematology 1995;55(2):93‐6. - PubMed
Dickgreber 2009 {published data only}
-
- Dickgreber N, Nagel S, Roscher K, Schuette W. Randomised, placebo‐controlled phase III trial of docetaxel plus carboplatin with or without levofloxacin prophylaxis in elderly patients with advanced non‐small cell lung cancer: The APRONTA Trial. [19th European Congress of Clinical Microbiology and Infectious Diseases]. Clinical Microbiology and Infection 2009;15:S519. - PubMed
Doorduijn 2005 {published data only}
-
- Doorduijn J, Buijt I, Holt B, Steijaert M, Uyl‐de Groot C, Sonneveld P. Self‐reported quality of life in elderly patients with aggressive non‐Hodgkin's lymphoma treated with CHOP chemotherapy. European Journal of Haematology 2005;75(2):116‐23. - PubMed
Dunlop 1996 {published data only}
-
- Dunlop DJ, Andersen S, Paul J, Soukop M, Ellis D, Tansey P. Filgrastim as an adjunct to combination chemotherapy in Hodgkin's disease. Annals of Oncology 1996:51. - PubMed
Eleutherakis‐Papaiakovou 2010 {published data only}
-
- Eleutherakis‐Papaiakovou E, Kostis E, Migkou M, Christoulas D, Terpos E, Gavriatopoulou M, et al. Prophylactic antibiotics for the prevention of neutropenic fever in patients undergoing autologous stem‐cell transplantation: results of a single institution, randomized phase 2 trial. American Journal of Hematology 2010;85(11):863‐7. - PubMed
Ernst 2008 {published data only}
-
- Ernst P, Bacigalupo A, Ringden O, Ruutu T, Kolb HJ, Lawrinson S, et al. A phase 3, randomized, placebo‐controlled trial of filgrastim in patients with haematological malignancies undergoing matched‐related allogeneic bone marrow transplantation. Archives of Drug Information 2008;1(3):89‐96. - PMC - PubMed
Faber 2006 {published data only}
-
- Faber E, Pytlik R, Slaby J, Zapletalova J, Kozak T, Raida L, et al. Individually determined dosing of filgrastim after autologous peripheral stem cell transplantation in patients with malignant lymphoma‐‐results of a prospective multicentre controlled trial. European Journal of Haematology 2006;77(6):493‐500. - PubMed
Feng 2014 {published data only}
-
- Feng X, Ruan Y, He Y, Zhang Y, Wu X, Liu H, et al. Prophylactic first‐line antibiotics reduce infectious fever and shorten hospital stay during chemotherapy‐induced agranulocytosis in childhood acute myeloid leukemia. Acta Haematologica 2014;132(1):112‐7. - PubMed
Fridrik 1997 {published data only}
-
- Fridrik MA, Greil R, Hausmaninger H, Krieger O, Oppitz P, Stoger M, et al. Randomized open label phase III trial of CEOP/IMVP‐Dexa alternating chemotherapy and filgrastim versus CEOP/IMVP‐Dexa alternating chemotherapy for aggressive non‐Hodgkin's lymphoma (NHL). A multicenter trial by the Austrian Working Group for Medical Tumor Therapy.[Erratum appears in Ann Hematol. 2014 Mar;93(3):539‐40]. Annals of Hematology 1997;75(4):135‐40. - PubMed
Garcia 2000 {published data only}
-
- Garcia G. Immediate vs. delayed imipenem treatment in cancer patients with profound neutropenia induced by high‐dose chemotherapy: Results of a randomized study. Revista Espanola de Quimioterapia 2000; Vol. 15:257‐63. - PubMed
Garcia‐Saenz 2002 {published data only}
-
- Garcia‐Saenz JA, Martin M, Casado A, Perez‐Segura P, Manrique I, Flores L, et al. Immediate vs. delayed imipenem treatment in cancer patients with profound neutropenia induced by high‐dose chemotherapy: results of a randomized study. Revista Espanola de Quimioterapia 2002;15(3):257‐63. - PubMed
Geissler 1997 {published data only}
-
- Geissler K, Koller E, Hubmann E, Niederwieser D, Hinterberger W, Geissler D, et al. Granulocyte colony‐stimulating factor as an adjunct to induction chemotherapy for adult acute lymphoblastic leukemia‐‐a randomized phase‐III study. Blood 1997;90(2):590‐6. - PubMed
Godwin 1998 {published data only}
-
- Godwin JE, Kopecky KJ, Head DR, Willman CL, Leith CP, Hynes HE, et al. A double‐blind placebo‐controlled trial of granulocyte colony‐stimulating factor in elderly patients with previously untreated acute myeloid leukemia: a Southwest oncology group study (9031). Blood 1998;91(10):3607‐15. - PubMed
Gonzalez‐Vicent 2004 {published data only}
-
- Gonzalez‐Vicent M, Madero L, Sevilla J, Ramirez M, Diaz MA. A prospective randomized study of clinical and economic consequences of using G‐CSF following autologous peripheral blood progenitor cell (PBPC) transplantation in children. Bone Marrow Transplantation 2004;34(12):1077‐81. - PubMed
Greenberg 1996 {published data only}
-
- Greenberg P, Advani R, Keating A, Gulati SC, Nimer S, Champlin R, et al. GM‐CSF accelerates neutrophil recovery after autologous hematopoietic stem cell transplantation. Bone Marrow Transplantation 1996;18(6):1057‐64. - PubMed
Gulati 1992 {published data only}
-
- Gulati SC, Bennett CL. Granulocyte‐macrophage colony‐stimulating factor (GM‐CSF) as adjunct therapy in relapsed Hodgkin disease. Annals of Internal Medicine 1992;116(3):177‐82. - PubMed
Hartmann 1997 {published data only}
-
- Hartmann LC, Tschetter LK, Habermann TM, Ebbert LP, Johnson PS, Mailliard JA, et al. Granulocyte colony‐stimulating factor in severe chemotherapy‐induced afebrile neutropenia. New England Journal of Medicine 1997;336(25):1776‐80. - PubMed
Heath 2003 {published data only}
-
- Heath JA, Steinherz PG, Altman A, Sather H, Jhanwar S, Halpern S, et al. Human granulocyte colony‐stimulating factor in children with high‐risk acute lymphoblastic leukemia: a Children's Cancer Group Study. Journal of Clinical Oncology 2003;21(8):1612‐7. - PubMed
Hecht 2010 {published data only}
-
- Hecht JR, Pillai M, Gollard R, Heim W, Swan F, Patel R, et al. A randomized, placebo‐controlled phase II study evaluating the reduction of neutropenia and febrile neutropenia in patients with colorectal cancer receiving pegfilgrastim with every‐2‐week chemotherapy. Clinical Colorectal Cancer 2010;9(2):95‐101. - PubMed
Heil 1997 {published data only}
-
- Heil G, Hoelzer D, Sanz MA, Lechner K, Liu Yin JA, Papa G, et al. A randomized, double‐blind, placebo‐controlled, phase III study of filgrastim in remission induction and consolidation therapy for adults with de novo acute myeloid leukemia. The International Acute Myeloid Leukemia Study Group. Blood 1997;90(12):4710‐8. - PubMed
Holowiecki 2002 {published data only}
-
- Holowiecki J, Giebel S, Krzemien S, Krawczyk‐Kulis M, Jagoda K, Kopera M, et al. G‐CSF administered in time‐sequenced setting during remission induction and consolidation therapy of adult acute lymphoblastic leukemia has beneficial influence on early recovery and possibly improves long‐term outcome: a randomized multicenter study. Leukemia & Lymphoma 2002;43(2):315‐25. - PubMed
Jones 1996 {published data only}
-
- Jones SE, Schottstaedt MW, Duncan LA, Kirby RL, Good RH, Mennel RG, et al. Randomized double‐blind prospective trial to evaluate the effects of sargramostim versus placebo in a moderate‐dose fluorouracil, doxorubicin, and cyclophosphamide adjuvant chemotherapy program for stage II and III breast cancer. Journal of Clinical Oncology 1996;14(11):2976‐83. - PubMed
Joshi 2003 {published data only}
-
- Joshi SS, Bishop MR, Lynch JC, Tarantolo SR, Abhyankar S, Bierman PJ, et al. Immunological and clinical effects of post‐transplant G‐CSF versus placebo in T‐cell replete allogeneic blood transplant patients: results from a randomized double‐blind study. Cytotherapy 2003;5(6):542‐52. - PubMed
Karp 1986 {published data only}
-
- Karp JE, Merz WG, Hendricksen C, Laughon B, Redden T, Bamberger BJ, et al. Infection management during antileukemia treatment‐induced granulocytopenia: the role for oral norfloxacin prophylaxis against infections arising from the gastrointestinal tract. Scandinavian Journal of Infectious Diseases Supplement 1986;48:66‐78. - PubMed
Kim 2005 {published data only}
-
- Kim S, Cho YH, Ko OB, Koo JE, Lee D, Chong YP, et al. Beneficial prophylactic antimicrobial use against infectious complications during autologous stem cell transplantation: a result of randomized phase II study in multiple myeloma and non‐Hodgkin's lymphoma patients. Blood. 2005; Vol. 11.
Kosaka 2015 {published data only}
-
- Kosaka Y, Rai Y, Masuda N, Takano T, Saeki T, Nakamura S, et al. Phase III placebo‐controlled, double‐blind, randomized trial of pegfilgrastim to reduce the risk of febrile neutropenia in breast cancer patients receiving docetaxel/cyclophosphamide chemotherapy. Support Care Cancer 2015;23(4):1137‐43. - PMC - PubMed
Ladenstein 2010 {published data only}
-
- Ladenstein R, Valteau‐Couanet D, Brock P, Yaniv I, Castel V, Laureys G, et al. Randomized Trial of prophylactic granulocyte colony‐stimulating factor during rapid COJEC induction in pediatric patients with high‐risk neuroblastoma: the European HR‐NBL1/SIOPEN study. Journal of Clinical Oncology 2010;28(21):3516‐24. - PubMed
Lalami 2004 {published data only}
-
- Lalami Y, Paesmans M, Aoun M, Munoz‐Bermeo R, Reuss K, Cherifi S, et al. A prospective randomised evaluation of G‐CSF or G‐CSF plus oral antibiotics in chemotherapy‐treated patients at high risk of developing febrile neutropenia. Supportive Care in Cancer 2004;12(10):725‐30. - PubMed
Lamy 1993 {published data only}
-
- Lamy T, Michelet C, Dauriac C, Grulois I, Donio PY, Prisé PY. Benefit of prophylaxis by intravenous systemic vancomycin in granulocytopenic patients: a prospective, randomized trial among 59 patients. Acta Haematologica 1993;90(3):109‐13. - PubMed
Larson 1998 {published data only}
-
- Larson RA, Dodge RK, Linker CA, Stone RM, Powell BL, Lee EJ, et al. A randomized controlled trial of filgrastim during remission induction and consolidation chemotherapy for adults with acute lymphoblastic leukemia: CALGB study 9111. Blood 1998;92(5):1556‐64. - PubMed
Lee 1998 {published data only}
-
- Lee SM, Radford JA, Dobson L, Huq T, Ryder WD, Pettengell Retal. Recombinant human granulocyte colony‐stimulating factor (filgrastim) following high‐dose chemotherapy and peripheral blood progenitor cell rescue in high‐grade non‐Hodgkin's lymphoma: clinical benefits at no extra cost. British Journal of Cancer 1998;77(8):1294‐9. - PMC - PubMed
Lee 2002 {published data only}
Lehrnbecher 2007 {published data only}
-
- Lehrnbecher T, Zimmermann M, Reinhardt D, Dworzak M, Stary J, Creutzig U. Prophylactic human granulocyte colony‐stimulating factor after induction therapy in pediatric acute myeloid leukemia. Blood 2007;109(3):936‐43. - PubMed
Little 2002 {published data only}
-
- Little MA, Morland B, Chisholm J, Hole A, Shankar A, Devine T, et al. A randomised study of prophylactic G‐CSF following MRC UKALL XI intensification regimen in childhood ALL and T‐NHL. Medical & Pediatric Oncology 2002;38(2):98‐103. - PubMed
Maiche 1993 {published data only}
-
- Maiche AG, Muhonen T. Granulocyte colony‐stimulating factor (G‐CSF) with or without a quinolone in the prevention of infection in cancer patients. European Journal of Cancer 1993;29A(10):1403‐5. - PubMed
McQuaker 1997 {published data only}
-
- McQuaker IG, Hunter AE, Pacey S, Haynes AP, Iqbal A, Russell NH. Low‐dose filgrastim significantly enhances neutrophil recovery following autologous peripheral‐blood stem‐cell transplantation in patients with lymphoproliferative disorders: evidence for clinical and economic benefit (Structured abstract). Journal of Clinical Oncology 1997;15(2):451‐7. - PubMed
Michel 2000 {published data only}
-
- Michel G, Landman‐Parker J, Auclerc MF, Mathey C, Leblanc T, Legall E, et al. Use of recombinant human granulocyte colony‐stimulating factor to increase chemotherapy dose‐intensity: a randomized trial in very high‐risk childhood acute lymphoblastic leukemia. Journal of Clinical Oncology 2000;18(7):1517‐24. - PubMed
Michon 1998 {published data only}
-
- Michon JM, Hartmann O, Bouffet E, Meresse V, Coze C, Rubie H, et al. An open‐label, multicentre, randomised phase 2 study of recombinant human granulocyte colony‐stimulating factor (filgrastim) as an adjunct to combination chemotherapy in paediatric patients with metastatic neuroblastoma. European Journal of Cancer 1998;34(7):1063‐9. - PubMed
Miles 1994 {published data only}
-
- Miles DW, Fogarty O, Ash CM, Rudd RM, Trask CW, Spiro SG, et al. Received dose‐intensity: a randomized trial of weekly chemotherapy with and without granulocyte colony‐stimulating factor in small‐cell lung cancer. Journal of Clinical Oncology 1994;12(1):77‐82. - PubMed
Nemunaitis 1995 {published data only}
-
- Nemunaitis J, Rosenfeld CS, Ash R, Freedman MH, Deeg HJ, Appelbaum F, et al. Phase III randomized, double‐blind placebo‐controlled trial of rhGM‐CSF following allogeneic bone marrow transplantation. Bone Marrow Transplantation 1995;15(6):949‐54. - PubMed
Nolan 2007 {published data only}
-
- Nolan L, Lorigan P, Chilton S, Newman J, Else R, Smith P, et al. Low‐dose lenograstim is as effective as standard dose in shortening neutrophil engraftment time following myeloablative chemotherapy and peripheral blood progenitor cell rescue. British Journal of Haematology 2007;137(5):436‐42. - PubMed
Ojeda 1999 {published data only}
-
- Ojeda E, Garcia‐Bustos J, Aguado M, Arrieta R, Quevedo E, Yuste VJ, et al. A prospective randomized trial of granulocyte colony‐stimulating factor therapy after autologous blood stem cell transplantation in adults. Bone Marrow Transplantation 1999;24(6):601‐7. - PubMed
Osby 2003 {published data only}
-
- Osby E, Hagberg H, Kvaløy S, Teerenhovi L, Anderson H, Cavallin‐Stahl E, et al. CHOP is superior to CNOP in elderly patients with aggressive lymphoma while outcome is unaffected by filgrastim treatment: results of a Nordic Lymphoma Group randomized trial. Blood 2003;101(10):3840‐8. - PubMed
Ottmann 1995 {published data only}
-
- Ottmann OG, Hoelzer D, Gracien E, Ganser A, Kelly K, Reutzel R, et al. Concomitant granulocyte colony‐stimulating factor and induction chemoradiotherapy in adult acute lymphoblastic leukemia: a randomized phase III trial. Blood 1995;86(2):444‐50. - PubMed
Patte 2002 {published data only}
-
- Patte C, Laplanche A, Bertozzi AI, Baruchel A, Frappaz D, Schmitt C, et al. Granulocyte colony‐stimulating factor in induction treatment of children with non‐Hodgkin's lymphoma: a randomized study of the French Society of Pediatric Oncology. Journal of Clinical Oncology 2002;20(2):441‐8. - PubMed
Petersen 1988 {published data only}
-
- Petersen F, Thornquist M, Buckner C, Counts G, Nelson N, Meyers J, et al. The effects of infection prevention regimens on early infectious complications in marrow transplant patients: a four arm randomized study. Infection 1988;16(4):199‐208. - PubMed
Pettengell 1992 {published data only}
-
- Pettengell R, Gurney H, Radford JA, Deakin DP, James R, Wilkinson PM, et al. Granulocyte colony‐stimulating factor to prevent dose‐limiting neutropenia in non‐Hodgkin's lymphoma: a randomized controlled trial. Blood 1992;80(6):1430‐6. - PubMed
Piccirillo 1999 {published data only}
-
- Piccirillo N, Sica S, Laurenti L, Chiusolo P, Barbera EO, Sora F, et al. Optimal timing of G‐CSF administration after CD34+ immunoselected peripheral blood progenitor cell transplantation. Bone Marrow Transplantation 1999;23(12):1245‐50. - PubMed
Pignon 1990 {published data only}
-
- Pignon B, Thiriet L, Aubert D, Pennaforte JL, Vilque JP, Lartigue B, et al. Evaluation of the efficacy of prophylactic intravenous antibiotherapy with ceftriaxone in post‐chemotherapy agranulocytic patients. Nouvelle Revue Française d'Hématologie 1990;32(4):249‐52. - PubMed
Przepiorka 2001 {published data only}
-
- Przepiorka D, Smith TL, Folloder J, Anderlini P, Chan KW, Korbling M, et al. Controlled trial of filgrastim for acceleration of neutrophil recovery after allogeneic blood stem cell transplantation from human leukocyte antigen‐matched related donors. Blood 2001;97(11):3405‐10. - PubMed
Pui 1997 {published data only}
-
- Pui CH, Boyett JM, Hughes WT, Rivera GK, Hancock ML, Sandlund JT, et al. Human granulocyte colony‐stimulating factor after induction chemotherapy in children with acute lymphoblastic leukemia. New England Journal of Medicine 1997;336(25):1781‐7. - PubMed
Rafecas 1989 {published data only}
-
- Rafecas FJ, Gil E, Martín G, Martínez J, Sanz G, Sempere A, et al. Oral ciprofloxacin in the prophylaxis of bacterial infection in neutropenic patients. A randomized, double‐blind, comparative clinical study. Revista Española de Quimioterapia 1989;2:174‐7.
Romieu 2007 {published data only}
-
- Romieu G, Clemens M, Mahlberg R, Fargeot P, Constenla M, Schutte M, et al. Pegfilgrastim supports delivery of FEC‐100 chemotherapy in elderly patients with high risk breast cancer: a randomized phase 2 trial. Critical Reviews in Oncology/hematology 2007;64(1):64‐72. - PubMed
Schmitz 2004 {published data only}
-
- Schmitz N, Ljungman P, Cordonnier C, Kempf C, Linkesch W, Alegre A, et al. Lenograstim after autologous peripheral blood progenitor cell transplantation: results of a double‐blind, randomized trial. Bone Marrow Transplantation 2004;34(11):955‐62. - PubMed
Schuette 2011 {published data only}
-
- Schuette W, Nagel S, Weikersthal LF, Pabst S, Schumann C, Deuss B, et al. Randomized phase III trial of docetaxel plus carboplatin with or without levofloxacin prophylaxis in elderly patients with advanced non‐small cell lung cancer: the APRONTA trial. Journal of Thoracic Oncology 2011;6(12):2090‐6. - PubMed
Seymour 1995 {published data only}
-
- Seymour AM, Campos E, Thatcher N, Greve J, Cunningham D, Howell A, et al. A single‐blind, randomised, vehicle‐controlled dose‐finding study of recombinant human granulocyte colony‐stimulating factor (lenograstim) in patients undergoing chemotherapy for solid cancers and lymphoma. European Journal of Cancer 1995;31A(13‐14):2157‐63. - PubMed
Spitzer 1994 {published data only}
-
- Spitzer G, Adkins DR, Spencer V, Dunphy FR, Petruska PJ, Velasquez WS, et al. Randomized study of growth factors post‐peripheral‐blood stem‐cell transplant: neutrophil recovery is improved with modest clinical benefit. Journal of Clinical Oncology 1994;12(4):661‐70. - PubMed
Stahel 1994 {published data only}
-
- Stahel RA, Jost LM, Cerny T, Pichert G, Honegger H, Tobler A, et al. Randomized study of recombinant human granulocyte colony‐stimulating factor after high‐dose chemotherapy and autologous bone marrow transplantation for high‐risk lymphoid malignancies. Journal of Clinical Oncology 1994;12(9):1931‐8. - PubMed
Suh 2008 {published data only}
-
- Suh Y, Chun C, Oh S. Prevention of infection after TAC chemotherapy for node‐positive breast cancer as an adjuvant therapy with or without ciprofloxacin. Annals of Oncology 2008;26(15S):81.
Talbot 1993 {published data only}
Timmer‐Bonte 2005 {published data only}
-
- Timmer‐Bonte JN, Boo TM, Smit HJ, Biesma B, Wilschut FA, Cheragwandi SA, et al. Prevention of chemotherapy‐induced febrile neutropenia by prophylactic antibiotics plus or minus granulocyte colony‐stimulating factor in small‐cell lung cancer: a Dutch Randomized Phase III Study. Journal of Clinical Oncology 2005;23(31):7974‐84. - PubMed
Tjan‐Heijnen 2003 {published data only}
-
- Tjan‐Heijnen VC, Caleo S, Postmus PE, Ardizzoni A, Burghouts JT, Buccholz E, et al. Economic evaluation of antibiotic prophylaxis in small‐cell lung cancer patients receiving chemotherapy: an EORTC double‐blind placebo‐controlled phase III study (08923). Annals of Oncology 2003;14(2):248‐57. - PubMed
Trigg 2000 {published data only}
-
- Trigg ME, Peters C, Zimmerman MB. Administration of recombinant human granulocyte‐macrophage colony‐stimulating factor to children undergoing allogeneic marrow transplantation: a prospective, randomized, double‐masked, placebo‐controlled trial. Pediatric Transplantation 2000;4(2):123‐31. - PubMed
Trillet‐Lenoir 1993 {published data only}
-
- Trillet‐Lenoir V, Green J, Manegold C, Pawel J, Gatzemeier U, Lebeau B, et al. Recombinant granulocyte colony stimulating factor reduces the infectious complications of cytotoxic chemotherapy. European Journal of Cancer 1993;29A(3):319‐24. - PubMed
Veyret 2006 {published data only}
-
- Veyret C, Levy C, Chollet P, Merrouche Y, Roche H, Kerbrat P, et al. Inflammatory breast cancer outcome with epirubicin‐based induction and maintenance chemotherapy: ten‐year results from the French Adjuvant Study Group GETIS 02 Trial. Cancer 2006;107(11):2535‐44. - PubMed
Vogel 2005 {published data only}
-
- Vogel CL, Wojtukiewicz MZ, Carroll RR, Tjulandin SA, Barajas‐Figueroa LJ, Wiens BL, et al. First and subsequent cycle use of pegfilgrastim prevents febrile neutropenia in patients with breast cancer: a multicenter, double‐blind, placebo‐controlled phase III study. Journal of Clinical Oncology 2005;23(6):1178‐84. - PubMed
Welte 1996 {published data only}
-
- Welte K, Reiter A, Mempel K, Pfetsch M, Schwab G, Schrappe M, et al. A randomized phase‐III study of the efficacy of granulocyte colony‐stimulating factor in children with high‐risk acute lymphoblastic leukemia. Berlin‐Frankfurt‐Munster Study Group. Blood 1996;87(8):3143‐50. - PubMed
Witz 1998 {published data only}
-
- Witz F, Sadoun A, Perrin MC, Berthou C, Briere J, Cahn JY, et al. A placebo‐controlled study of recombinant human granulocyte‐macrophage colony‐stimulating factor administered during and after induction treatment for de novo acute myelogenous leukemia in elderly patients. Groupe Ouest Est Leucemies Aigues Myeloblastiques (GOELAM). Blood 1998;91(8):2722‐30. - PubMed
Yamada 1993 {published data only}
-
- Yamada T, Dan K, Nomura T. Prevention of bacterial and fungal infections in acute leukemia patients: a new and potent combination of oral norfloxacin and amphotericin B. Internal Medicine (Tokyo, Japan) 1993;32(9):710‐5. - PubMed
Yau 1996 {published data only}
-
- Yau JC, Neidhart JA, Triozzi P, Verma S, Nemunaitis J, Quick DP, et al. Randomized placebo‐controlled trial of granulocyte‐macrophage colony‐stimulating‐factor support for dose‐intensive cyclophosphamide, etoposide, and cisplatin. American Journal of Hematology 1996;51(4):289‐95. - PubMed
Additional references
Alvarado 1999
-
- Alvarado Ibarra ML, Borbolla Escoboza JR, Lopez‐Hernandez MA, Gonzalez‐Avante CM, FloresChapa JD, Trueba Christy E, et al. Neutrophil recovery time and adverse side effects in acute leukemia patients treated with intensive chemotherapy and concomitant G or GM‐CSF. Revista de Investigacion Clinica 1999;51(2):77‐80. [PUBMED: 10410585] - PubMed
Beveridge 1997
-
- Beveridge RA, Miller JA, Kales AN, Binder RA, Robert NJ, Heisrath‐Evans J, et al. Randomized trial comparing the tolerability of sargramostim (yeast‐derived RhuGM‐CSF) and filgrastim (bacteria‐derived RhuG‐CSF) in cancer patients receiving myelosuppressive chemotherapy. Supportive Care in Cancer 1997;5(4):289‐98. [PUBMED: 9257425] - PubMed
Beveridge 1998
-
- Beveridge RA, Miller JA, Kales AN, Binder RA, Robert NJ, Harvey JH, et al. A comparison of efficacy of sargramostim (yeast‐derived RhuGM‐CSF) and filgrastim (bacteria‐derived RhuG‐CSF) in the therapeutic setting of chemotherapy‐induced myelosuppression. Cancer Investigation 1998;16(6):366‐73. [PUBMED: 9679526] - PubMed
Bodey 1966
-
- Bodey GP, Buckley M, Sathe YS, Freireich EJ. Quantitative relationships between circulating leukocytes and infection in patients with acute leukemia. Annals of Internal Medicine 1966;64(2):328‐40. - PubMed
Bodey 1986
-
- Bodey GP. Infection in cancer patients. A continuing association. American Journal of Medicine 1986;81(1A):11‐26. - PubMed
Bohlius 2008
Bronchud 1988
Bucaneve 2005
-
- Bucaneve G, Micozzi A, Menichetti F, Martino P, Dionisi MS, Martinelli G, et al. Levofloxacin to prevent bacterial infection in patients with cancer and neutropenia. New England Journal of Medicine 2005;353(10):977‐87. - PubMed
Carratala 1995
-
- Carratala J, Fernandez‐Sevilla A, Tubau F, Callis M, Gudiol F. Emergence of quinolone‐resistant Escherichia coli bacteremia in neutropenic patients with cancer who have received prophylactic norfloxacin. Clinical Infectious Diseases 1995;20(3):557‐60. - PubMed
Clark 2005
-
- Clark OA, Lyman GH, Castro AA, Clark LG, Djulbegovic B. Colony‐stimulating factors for chemotherapy‐induced febrile neutropenia: a meta‐analysis of randomized controlled trials. Journal of Clinical Oncology 2005;23(18):4198‐214. - PubMed
Crawford 1991
-
- Crawford J, Ozer H, Stoller R, Johnson D, Lyman G, Tabbara I, et al. Reduction by granulocyte colony‐stimulating factor of fever and neutropenia induced by chemotherapy in patients with small‐cell lung cancer. New England Journal of Medicine 1991;325(3):164‐70. - PubMed
Cruciani 2003
-
- Cruciani M, Malena M, Bosco O, Nardi S, Serpelloni G, Mengoli C. Reappraisal with meta‐analysis of the addition of Gram‐positive prophylaxis to fluoroquinolone in neutropenic patients. Journal of Clinical Oncology 2003;21(22):4127‐37. - PubMed
Deeks 2011
-
- Deeks JJ, Higgins JPT, Altman DG. Chapter 9: Analysing data and undertaking meta‐analyses. In: Higgins JPT, Green S editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011].The Cochrane Collaboration. Available from www.cochrane‐handbook.org. 2011.
Engels 1998
-
- Engels EA, Lau J, Barza M. Efficacy of quinolone prophylaxis in neutropenic cancer patients: a meta‐analysis. Journal of Clinical Oncology 1998;16(3):1179‐87. - PubMed
Fischmeister 1999
-
- Fischmeister G, Kurz M, Haas OA, Micksche M, Buchinger P, Printz D, et al. G‐CSF versus GM‐CSF for stimulation of peripheral blood progenitor cells (PBPC) and leukocytes in healthy volunteers: comparison of efficacy and tolerability. Annals of Hematology 1999;78(3):117‐23. [PUBMED: 10211753] - PubMed
Gafter‐Gvili 2005
-
- Gafter‐Gvili A, Fraser A, Paul M, Leibovici L. Meta‐Analysis: Antibiotic prophylaxis reduces mortality in neutropenic patients. Annals of Internal Medicine 2005;142(12 Pt 1):979‐95. - PubMed
Gafter‐Gvili 2007
-
- Gafter‐Gvili A, Paul M, Fraser A, Leibovici L. Effect of quinolone prophylaxis in afebrile neutropenic patients on microbial resistance: systematic review and meta‐analysis. Journal of Antimicrobial Chemotherapy 2007;59(1):5‐22. - PubMed
Gafter‐Gvili 2012
Griffin 1990
-
- Griffin JD, Cannistra SA, Sullivan R, Demetri GD, Ernst TJ, Kanakura Y. The biology of GM‐CSF: regulation of production and interaction with its receptor. International Journal of Cell Cloning 1990;8 Suppl 1:35‐44. - PubMed
Hackshaw 2004
Higgins 2011
-
- Higgins JPT, Deeks JJ (editors). Chapter 7: Selecting studies and collecting data. In: Higgins JPT, Green S editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration. Available from www.cochrane‐handbook.org. 2011.
Higgins 2011a
-
- Higgins JPT, Altman DG. Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Green S editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration. Available from www.cochrane‐handbook.org. 2011.
Higgins 2011b
-
- Higgins JPT, Deeks JJ, Altman DG (editors). Chapter 16: Special topics in statistics. In: Higgins JPT, Green S editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011].The Cochrane Collaboration. Available from www.cochrane‐handbook.org. 2011.
Holmes 2002
-
- Holmes FA, Jones SE, O'Shaughnessy J, Vukelja S, George T, Savin M, et al. Comparable efficacy and safety profiles of once‐per‐cycle pegfilgrastim and daily injection filgrastim in chemotherapy‐induced neutropenia: a multicenter dose‐finding study in women with breast cancer. Annals of Oncology 2002;13(6):903‐9. - PubMed
Hovgaard 1992
-
- Hovgaard DJ, Nissen NI. Effect of recombinant human granulocyte‐macrophage colony‐stimulating factor in patients with Hodgkin's disease: a phase I/II study. Journal of Clinical Oncology 1992;10(3):390‐7. [PUBMED: 1740678] - PubMed
Johnston 2000
-
- Johnston E, Crawford J, Blackwell S, Bjurstrom T, Lockbaum P, Roskos L, et al. Randomized, dose‐escalation study of SD/01 compared with daily filgrastim in patients receiving chemotherapy. Journal of Clinical Oncology 2000;18(13):2522‐8. - PubMed
Karp 1987
-
- Karp JE, Merz WG, Hendricksen C, Laughon B, Redden T, Bamberger BJ, et al. Oral norfloxacin for prevention of gram‐negative bacterial infections in patients with acute leukemia and granulocytopenia. A randomized, double‐blind, placebo‐controlled trial. Annals of Internal Medicine 1987;106(1):1‐7. - PubMed
Kuderer 2006
-
- Kuderer NM, Dale DC, Crawford J, Cosler LE, Lyman GH. Mortality, morbidity, and cost associated with febrile neutropenia in adult cancer patients. Cancer 2006;106(10):2258‐66. - PubMed
Lefebvre 2011
-
- Lefebvre C, Manheimer E, Glanville J. Chapter 6: Searching for studies. In: Higgins JPT, Green S editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, Available from www.cochrane‐handbook.org. 2011.
Leibovici 2006
-
- Leibovici L, Paul M, Cullen M, Bucaneve G, Gafter‐Gvili A, Fraser A, et al. Antibiotic prophylaxis in neutropenic patients: new evidence, practical decisions. Cancer 2006;107(8):1743‐51. - PubMed
Lew 1995
-
- Lew MA, Kehoe K, Ritz J, Antman KH, Nadler L, Kalish LA, et al. Ciprofloxacin versus trimethoprim/sulfamethoxazole for prophylaxis of bacterial infections in bone marrow transplant recipients: a randomized, controlled trial. Journal of Clinical Oncology 1995;13(1):239‐50. - PubMed
Lyman 2002
-
- Lyman GH, Kuderer NM, Djulbegovic B. Prophylactic granulocyte colony‐stimulating factor in patients receiving dose‐intensive cancer chemotherapy: A meta‐analysis. American Journal of Medicine 2002;112(5):406‐11. - PubMed
Lyman 2010
-
- Lyman GH, Dale DC, Wolff DA, Culakova E, Poniewierski MS, Kuderer NM, et al. Acute myeloid leukemia or myelodysplastic syndrome in randomized controlled clinical trials of cancer chemotherapy with granulocyte colony‐stimulating factor: a systematic review. Journal of Clinical Oncology 2010;28(17):2914‐24. [PUBMED: 20385991] - PubMed
Moher 2009
-
- Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta‐Analyses: the PRISMA statement. Journal of Clinical Epidemiology 2009;62(10):1006‐12. [PUBMED: 19631508] - PubMed
Morstyn 1988
-
- Morstyn G, Campbell L, Souza LM, Alton NK, Keech J, Green M, et al. Effect of granulocyte colony stimulating factor on neutropenia induced by cytotoxic chemotherapy. Lancet 1988;1(8587):667‐72. - PubMed
Parmar 1998
-
- Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta‐analyses of the published literature for survival endpoints. Statistics in Medicine 1998;17(24):2815‐34. - PubMed
Pizzo 1999
-
- Pizzo PA. Fever in immunocompromised patients. New England Journal of Medicine 1999;341(12):893‐900. - PubMed
Review Manager (RevMan) [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Smith 2015
-
- Smith TJ, Bohlke K, Lyman GH, Carson KR, Crawford J, Cross SJ, et al. Recommendations for the use of WBC growth factors: American Society of Clinical Oncology Clinical Practice Guideline Update. Journal of Clinical Oncology 2015;33(28):3199‐212. - PubMed
Somolinos 1992
-
- Somolinos N, Arranz R, Rey MC, Jimenez ML. Superinfections by Escherichia coli resistant to fluoroquinolones in immunocompromised patients. Journal of Antimicrobial Chemotherapy 1992;30(5):730‐1. - PubMed
Sterne 2011
-
- Sterne JAC, Egger M, Moher D. Chapter 10: Addressing reporting biases. In: Higgins JPT, Green S editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, Available from www.cochrane‐handbook.org.. 2011.
Sung 2004
-
- Sung L, Nathan PC, Lange B, Beyene J, Buchanan JR. Prophylactic granulocyte colony‐stimulating factor and granulocyte‐macrophage colony‐stimulating factor decrease febrile neutropenia after chemotherapy in children with cancer: A meta‐analysis of randomized controlled trials. Journal of Clinical Oncology 2004;22(16):3350‐6. - PubMed
Sung 2007
-
- Sung L, Nathan PC, Alibhai SM, Tomlinson GA, Beyene J. Meta‐analysis: effect of prophylactic hematopoietic colony‐stimulating factors on mortality and outcomes of infection. Annals of Internal Medicine 2007;147(6):400‐11. - PubMed
Tierney 2007
Vehreschild 2014
-
- Vehreschild JJ, Bohme A, Cornely OA, Kahl C, Karthaus M, Kreuzer KA, et al. Prophylaxis of infectious complications with colony‐stimulating factors in adult cancer patients undergoing chemotherapy‐evidence‐based guidelines from the Infectious Diseases Working Party AGIHO of the German Society for Haematology and Medical Oncology (DGHO). Annals of Oncology 2014;25(9):1709‐18. [PUBMED: 24631945] - PubMed
Wittman 2006
-
- Wittman B, Horan J, Lyman GH. Prophylactic colony‐stimulating factors in children receiving myelosuppressive chemotherapy: a meta‐analysis of randomized controlled trials. Cancer Treatment Reviews 2006;32(4):289‐303. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous