Metal Chelation Modulates Phototherapeutic Properties of Mitoxantrone-Loaded Porphyrin-Phospholipid Liposomes
- PMID: 26691879
- PMCID: PMC6247800
- DOI: 10.1021/acs.molpharmaceut.5b00653
Metal Chelation Modulates Phototherapeutic Properties of Mitoxantrone-Loaded Porphyrin-Phospholipid Liposomes
Abstract
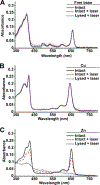
Liposomes incorporating porphyrin-phospholipid (PoP) can be formulated to release entrapped contents in response to near-infrared (NIR) laser irradiation. Here, we examine effects of chelating copper or zinc into the PoP. Cu(II) and Zn(II) PoP liposomes, containing 10 molar % HPPH-lipid, exhibited unique photophysical properties and released entrapped cargo in response to NIR light. Cu-PoP liposomes exhibited minimal fluorescence and reduced production of reactive oxygen species upon irradiation. Zn-PoP liposomes retained fluorescence and singlet oxygen generation properties; however, they rapidly self-bleached under laser irradiation. Compared to the free base form, both Cu- and Zn-PoP liposomes exhibited reduced phototoxicity in mice. When loaded with mitoxantrone and administered intravenously at 5 mg/kg to mice bearing human pancreatic cancer xenografts, synergistic effects between the drug and the light treatment (for this particular dose and formulation) were realized with metallo-PoP liposomes. The drug-light-interval affected chemophototherapy efficacy and safety.
Keywords: Chemophototherapy; light-triggered; liposomes; metalloporphyrins; mitoxantrone; photobleaching; porphyrin-phospholipid; tumor ablation.
Figures
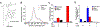

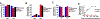
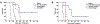
Similar articles
-
Rapid Light-Triggered Drug Release in Liposomes Containing Small Amounts of Unsaturated and Porphyrin-Phospholipids.Small. 2016 Jun;12(22):3039-47. doi: 10.1002/smll.201503966. Epub 2016 Apr 28. Small. 2016. PMID: 27121003 Free PMC article.
-
Vessel-Targeted Chemophototherapy with Cationic Porphyrin-Phospholipid Liposomes.Mol Cancer Ther. 2017 Nov;16(11):2452-2461. doi: 10.1158/1535-7163.MCT-17-0276. Epub 2017 Jul 20. Mol Cancer Ther. 2017. PMID: 28729400 Free PMC article.
-
Short Drug-Light Intervals Improve Liposomal Chemophototherapy in Mice Bearing MIA PaCa-2 Xenografts.Mol Pharm. 2018 Sep 4;15(9):3682-3689. doi: 10.1021/acs.molpharmaceut.8b00052. Epub 2018 Apr 2. Mol Pharm. 2018. PMID: 29608312
-
Chemophototherapeutic Ablation of Doxorubicin-Resistant Human Ovarian Tumor Cells.Photochem Photobiol. 2023 Mar;99(2):844-849. doi: 10.1111/php.13677. Epub 2022 Aug 2. Photochem Photobiol. 2023. PMID: 35842741 Free PMC article. Review.
-
Photo-triggerable liposomal drug delivery systems: from simple porphyrin insertion in the lipid bilayer towards supramolecular assemblies of lipid-porphyrin conjugates.J Mater Chem B. 2019 Mar 21;7(11):1805-1823. doi: 10.1039/c9tb00015a. Epub 2019 Feb 18. J Mater Chem B. 2019. PMID: 32255044 Review.
Cited by
-
Rapid Light-Triggered Drug Release in Liposomes Containing Small Amounts of Unsaturated and Porphyrin-Phospholipids.Small. 2016 Jun;12(22):3039-47. doi: 10.1002/smll.201503966. Epub 2016 Apr 28. Small. 2016. PMID: 27121003 Free PMC article.
-
Zwitterionic Cross-Linked Biodegradable Nanocapsules for Cancer Imaging.Langmuir. 2019 Feb 5;35(5):1440-1449. doi: 10.1021/acs.langmuir.8b01633. Epub 2018 Aug 21. Langmuir. 2019. PMID: 30086625 Free PMC article.
-
A dual-channel endoscope for quantitative imaging, monitoring, and triggering of doxorubicin release from liposomes in living mice.Sci Rep. 2017 Nov 14;7(1):15578. doi: 10.1038/s41598-017-15790-y. Sci Rep. 2017. PMID: 29138489 Free PMC article.
-
Fluence rate-dependent kinetics of light-triggered liposomal doxorubicin assessed by quantitative fluorescence-based endoscopic probe.Res Sq [Preprint]. 2025 Jan 3:rs.3.rs-5727534. doi: 10.21203/rs.3.rs-5727534/v1. Res Sq. 2025. Update in: Int J Mol Sci. 2025 Jan 30;26(3):1212. doi: 10.3390/ijms26031212. PMID: 39801514 Free PMC article. Updated. Preprint.
-
A toolbox for enzymatic modification of nucleic acids with photosensitizers for photodynamic therapy.RSC Chem Biol. 2024 Jul 8;5(9):841-852. doi: 10.1039/d4cb00103f. eCollection 2024 Aug 28. RSC Chem Biol. 2024. PMID: 39211468 Free PMC article.
References
-
- Allen TM; Cullis PR Liposomal Drug Delivery Systems: From Concept to Clinical Applications. Adv. Drug Deliv. Rev 2013, 65 (1), 36–48. - PubMed
-
- Barenholz Y Doxil® — The First FDA-Approved Nano-Drug: Lessons Learned. J. Controlled Release 2012, 160 (2), 117–134. - PubMed
-
- Kingsley JD; Dou H; Morehead J; Rabinow B; Gendelman HE; Destache CJ Nanotechnology: A Focus on Nanoparticles as a Drug Delivery System. J. Neuroimmune Pharmacol 2006, 1 (3), 340–350. - PubMed
-
- Allen TM; Cullis PR Drug Delivery Systems: Entering the Mainstream. Science 2004, 303 (5665), 1818–1822. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

