Tumor suppressor SPOP mediates the proteasomal degradation of progesterone receptors (PRs) in breast cancer cells
- PMID: 26693071
- PMCID: PMC4656742
Tumor suppressor SPOP mediates the proteasomal degradation of progesterone receptors (PRs) in breast cancer cells
Abstract
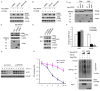
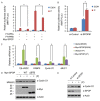
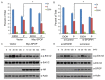
Progesterone induces proliferation of breast cancer cells and contributes to the development of breast cancer. The effects of progesterone are mediated by progesterone receptors (PRs). However, it is still not fully understood how the proliferative effects of PR is regulated in vivo. Increasing amount of evidence strongly suggests that dysregulation of ubiquitin-proteasome system is closely associated with cancer pathogenesis. Speckle-type POZ protein (SPOP) is an adaptor protein of the CUL3-based E3 ubiquitin ligase complexes. SPOP represents one of the highest loci for loss of heterozygosity (LOH) in breast cancer. SPOP downregulation contributes to breast cancer cell growth and invasion. In this study, we revealed PR as a bona fide substrate for SPOP. SPOP interacts with PR in vivo and targets PR for ubiquitin-dependent proteasomal degradation. Moreover, SPOP suppresses progesteroneinduced PR transactivation, S phase entry, and Erk1/2 activation. Our study revealed novel molecular mechanisms underlying the regulation of PR protein homeostasis in breast cancer cells, and provided insights in understanding the relationship between SPOP inactivation and the development of breast cancer.
Keywords: Breast cancer; SPOP; degradation; progesterone receptor; ubiquitination.
Figures

Similar articles
-
Endometrial cancer-associated mutants of SPOP are defective in regulating estrogen receptor-α protein turnover.Cell Death Dis. 2015 Mar 12;6(3):e1687. doi: 10.1038/cddis.2015.47. Cell Death Dis. 2015. PMID: 25766326 Free PMC article.
-
Destruction of DDIT3/CHOP protein by wild-type SPOP but not prostate cancer-associated mutants.Hum Mutat. 2014 Sep;35(9):1142-51. doi: 10.1002/humu.22614. Epub 2014 Jul 23. Hum Mutat. 2014. PMID: 24990631
-
SPOP promotes ATF2 ubiquitination and degradation to suppress prostate cancer progression.J Exp Clin Cancer Res. 2018 Jul 11;37(1):145. doi: 10.1186/s13046-018-0809-0. J Exp Clin Cancer Res. 2018. PMID: 29996942 Free PMC article.
-
The ubiquitin ligase adaptor SPOP in cancer.FEBS J. 2019 Oct;286(20):3946-3958. doi: 10.1111/febs.15056. Epub 2019 Sep 18. FEBS J. 2019. PMID: 31495053 Free PMC article. Review.
-
Novel insights into the SPOP E3 ubiquitin ligase: From the regulation of molecular mechanisms to tumorigenesis.Biomed Pharmacother. 2022 May;149:112882. doi: 10.1016/j.biopha.2022.112882. Epub 2022 Mar 29. Biomed Pharmacother. 2022. PMID: 35364375 Review.
Cited by
-
P38α MAPK is a gatekeeper of uterine progesterone responsiveness at peri-implantation via Ube3c-mediated PGR degradation.Proc Natl Acad Sci U S A. 2022 Aug 9;119(32):e2206000119. doi: 10.1073/pnas.2206000119. Epub 2022 Aug 1. Proc Natl Acad Sci U S A. 2022. PMID: 35914132 Free PMC article.
-
Chromatin-Bound Cullin-Ring Ligases: Regulatory Roles in DNA Replication and Potential Targeting for Cancer Therapy.Front Mol Biosci. 2018 Mar 13;5:19. doi: 10.3389/fmolb.2018.00019. eCollection 2018. Front Mol Biosci. 2018. PMID: 29594129 Free PMC article. Review.
-
E3 Ubiquitin Ligases in Breast Cancer Metastasis: A Systematic Review of Pathogenic Functions and Clinical Implications.Front Oncol. 2021 Oct 22;11:752604. doi: 10.3389/fonc.2021.752604. eCollection 2021. Front Oncol. 2021. PMID: 34745984 Free PMC article.
-
Uterine function in the mouse requires speckle-type poz protein.Biol Reprod. 2018 Jun 1;98(6):856-869. doi: 10.1093/biolre/ioy060. Biol Reprod. 2018. PMID: 29546395 Free PMC article.
-
Cancer Mutations of the Tumor Suppressor SPOP Disrupt the Formation of Active, Phase-Separated Compartments.Mol Cell. 2018 Oct 4;72(1):19-36.e8. doi: 10.1016/j.molcel.2018.08.027. Epub 2018 Sep 20. Mol Cell. 2018. PMID: 30244836 Free PMC article.
References
-
- Lanari C, Wargon V, Rojas P, Molinolo AA. Antiprogestins in breast cancer treatment: are we ready? Endocr Relat Cancer. 2012;19:R35–50. - PubMed
-
- Beato M, Herrlich P, Schutz G. Steroid hormone receptors: many actors in search of a plot. Cell. 1995;83:851–857. - PubMed
-
- McKenna NJ, Lanz RB, O’Malley BW. Nuclear receptor coregulators: cellular and molecular biology. Endocr Rev. 1999;20:321–344. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
