Epstein-Barr virus-induced gene 3 suppresses T helper type 1, type 17 and type 2 immune responses after Trypanosoma cruzi infection and inhibits parasite replication by interfering with alternative macrophage activation
- PMID: 26694585
- PMCID: PMC4754611
- DOI: 10.1111/imm.12565
Epstein-Barr virus-induced gene 3 suppresses T helper type 1, type 17 and type 2 immune responses after Trypanosoma cruzi infection and inhibits parasite replication by interfering with alternative macrophage activation
Abstract
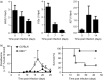
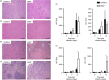
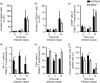
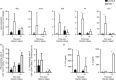
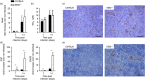
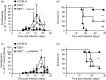
The Epstein-Barr virus-induced gene 3 (EBI3) is a member of the interleukin-12 (IL)-12) family structurally related to the subunit p40 of IL-12 and forms a heterodimer either with the p28 subunit to build IL-27 or with p35 to form IL-35. Interleukin-27 is secreted by antigen-presenting cells whereas IL-35 appears to be produced mainly by regulatory T cells and regulatory B cells but both cytokines negatively regulate inflammatory immune responses. We here analysed the function of EBI3 during infection with the intracellular parasite Trypanosoma cruzi. Compared with C57BL/6 wild-type mice, EBI3-deficient (EBI3(-/-) ) mice showed a higher parasitaemia associated with an increased mortality rate. The EBI3(-/-) mice displayed an elevated inflammatory immune response with an increased production of T helper type 1 (Th1-), Th2- and Th17-derived cytokines. The increased Th2 immune response appears to have over-ridden the otherwise protective Th1 and Th17 immune responses by the induction of arginase-1-expressing alternatively activated macrophages in these mice. Hence, neutralization of IL-4 and arginase-1 activity partially restored protective immune responses in EBI3(-/-) mice. So far, our results demonstrate that EBI3 is an essential general regulator of inflammatory immune responses in experimental Chagas disease and is required for control of T. cruzi infection by inhibiting Th2-dependent alternative macrophage activation. Further studies are needed to dissect the underlying mechanisms and clarify whether EBI3 association with IL-27 or/and IL-35 accounts for its anti-inflammatory character in parasitic disease.
Keywords: alternative macrophage activation; interleukin-27; interleukin-35; parasitic infection.
© 2015 John Wiley & Sons Ltd.
Figures
References
-
- World Health Organization : Chagas disease (American trypanosomiasis), Fact sheet No. 340. 2015.
-
- Erdmann H, Roßnagel C, Böhme J, Iwakura Y, Jacobs T, Schaible UE et al IL‐17A promotes macrophage effector mechanisms against Trypanosoma cruzi by trapping parasites in the endolysosomal compartment. Immunobiology 2013; 218:910–23. - PubMed
-
- Abrahamsohn IA, Coffman RL. Trypanosoma cruzi: IL‐10, TNF, IFN‐γ, and IL‐12 regulate innate and acquired immunity to infection. Exp Parasitol 1996; 84:231–44. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

